Purin: Gefahr Gicht & Harnsteine - einfach erklärt
Wie Sie über eine purinbewusste Ernährung Gicht und Harnsteinen entgegenwirken – oder Ihren Gesundheitszustand verbessern.
Inhaltsverzeichnis
Dieser Beitrag fasst das Wesentliche zusammen. Die vollständige Fassung mit Studiendetails und Therapiehinweisen bringt Ihnen dieser Link. Unsere Quellenangaben (75 Quellen), grösstenteils wissenschaftliche Studien, gelten für beide Beiträge.
Einleitung
Seit Jahrzehnten stellt die Ernährungswissenschaft einzelne Stoffe in den Mittelpunkt – oft zulasten ganzheitlicher Zusammenhänge. Erstaunlich unterrepräsentiert bleiben dabei die Purine, obwohl sie zu den Grundbausteinen des Lebens gehören. In Wahrheit erfüllen sie lebenswichtige Funktionen im zellulären Stoffwechsel.
Die Ernährungslehre diskutiert Purine praktisch nur im Zusammenhang mit Krankheiten, die aus ihrem Abbauprodukt entstehen – der Harnsäure. Problematisch wirken ein Übermass an purinreicher Nahrung – ebenso ein gestörter Abbau oder eine geringere Ausscheidung.17,57
Ein häufiger Grund für Verwirrungen liegt in der Verwechslung von Purinen mit Proteinen. Beide Stoffe erfüllen jedoch unterschiedliche Funktionen im menschlichen Organismus.
1 Was bedeuten Purine, Proteine und Aminosäuren?
Purine bilden das Grundgerüst für die Purinbasen Adenin und Guanin. Als Bestandteile universeller Energieträger, Signalmoleküle und Coenzyme steuern diese Purinbasen zentrale biochemische Prozesse der Zelle. Zudem dienen sie als Bausteine der Erbinformation (in DNA und RNA).1,3,56
Proteine hingegen gehören zu den Makronährstoffen und bestehen aus Aminosäuren. Sie übernehmen Aufbau und Erhalt von Gewebe – Muskeln, Haut, Organe. Daneben agieren sie als Enzyme für Stoffwechselreaktionen sowie als Hormone und Bestandteile des Immunsystems.8
Purine kommen häufig in proteinreichen Lebensmitteln vor.38 Deshalb entstehen oft Verwechslungen zwischen beiden Stoffgruppen. Bei ihrem Abbau spielen allerdings dieselben Organe eine wichtige Rolle: Leber und Nieren.
- Die Leber verstoffwechselt Purine zu Harnsäure und Aminosäuren (als Proteinbestandteile) zu Harnstoff. Beide Abbauprodukte scheidet der Körper über die Nieren aus.
- Purine verknüpfen den Zellstoffwechsel direkt mit Synthese und Abbau von Harnsäure – und damit mit Gicht, Harnsäuresteinen und verwandten Erkrankungen.
Warum kennt jeder Proteine, kaum jemand Purine?
Muskelaufbau und Leistungsfähigkeit zählen zu den sichtbaren Vorteilen einer ausreichenden Proteinzufuhr. Purine dagegen wirken im Verborgenen – u.a. als DNA- und RNA-Bausteine.1 Diese Funktionen wirken für Laien oft schwer zugänglich und bleiben deshalb unbeachtet.
Das Thema Purine taucht fast ausschliesslich im Zusammenhang mit Krankheiten wie Gicht oder Harnsäuresteinen auf. Dadurch erhielten diese Stoffe ein negatives Image. Denn ein chronischer Überschuss an Purinen führt in der Regel zu erhöhten Harnsäurespiegeln (Hyperurikämie bzw. Hyperurikosurie).59 Forschungsdetails zu Proteinen, Purinen und Pyrimidinen finden Sie im ClickFor beim ausführlichen Beitrag.3,4,5,6,7
Purine stellt der menschliche Körper zu ca. 2/3 selbst her und nimmt den Rest mit der Ernährung auf.59 Deshalb existiert keine "essenzielle Purinbase", die zwingend aus der Nahrung nötig ist. Diese Autarkie unterscheidet Purine grundlegend von den Aminosäuren.
2 Aufnahme, Nutzung und Abbau
Aufnahme: Neben der körpereigenen Synthese nimmt der Körper Purine als Bestandteile von Nukleinsäuren (DNA und RNA) über die Nahrung auf – primär im Dünndarm. Dort zerlegen Enzyme DNA und RNA in kleinere Abschnitte und spalten sie weiter zu Nukleotiden, Nukleosiden und freien Basen.9,10,42
Der Körper nimmt Purine fast vollständig ins Blut auf, selbst wenn er sie nicht sofort benötigt. Evolutiv ergibt das Sinn: In Zeiten von Nahrungsmangel war es günstiger, alles Wertvolle aufzunehmen, statt etwas ungenutzt auszuscheiden.
Nutzung: In der Leber baut der Körper damit DNA und RNA auf. Zudem bildet er daraus Energieträger (wie ATP und GTP) sowie Signalstoffe (wie cAMP). Zusätzlich recycelt der Körper Purine über den Salvage-Weg – er gewinnt aus Abbauprodukten von Nukleinsäuren wertvolle Moleküle zurück.2
Abbau: Sobald der Körper mehr Purine erhält, als er benötigt, erfolgt deren Abbau. Die Leber bildet daraus Harnsäure und gibt sie ins Blutplasma ab. Danach filtert die Niere die Harnsäure heraus und reguliert ihre Menge über Rückaufnahme und Ausscheidung. Insgesamt scheidet die Niere etwa zwei Drittel über den Urin aus, der Gastrointestinaltrakt ein Drittel.11,12,13,14
Wie Leber und Nieren gesund halten
Eine chronisch erhöhte Zufuhr von purin- und proteinreichen Lebensmitteln belastet den Stoffwechsel. Je mehr wir davon zuführen, desto stärker arbeiten Leber und Nieren. Diese Organe verfügen – anders als Muskeln – nur über begrenzte Regenerations- und Leistungsreserven. Daher führt eine eiweiss- und purinreiche Ernährung nicht zur Stärkung, sondern zur Abnutzung von Leber und Nieren.52 Dies ist nicht nur bei genetischer Vorbelastung oder Vorerkrankungen ein Problem, sondern auch für gesunde Menschen.
Während für die Proteinzufuhr minimale und maximale Grenzbereiche existieren, fehlen für den Purinkonsum vergleichbare Richtwerte. Aus offensichtlichen Gründen: Der Körper reguliert den Harnsäurespiegel über mehrere komplex verknüpfte Stoffwechselprozesse.
Wichtiger als der Versuch, die Nieren zu "reinigen", ist es, sie nicht zu überlasten. Also, den Purin- und Eiweisskonsum bewusst zu senken – und weder zu wenig noch zu viel Wasser aufzunehmen. Das heisst für Menschen mit chronischer Nierenerkrankung: Zu geringe (< 1 l/Tag) und zu hohe (> 2,5 l/Tag) Flüssigkeitsaufnahmen beschleunigen laut einer Kohortenstudie den Funktionsverlust. Für Gesunde gibt es bisher keine vergleichbaren Daten.15
Gesundes Darmmikrobiom unterstützt Harnsäureabbau
Gesunde Darmbakterien tragen wesentlich zum Abbau von Harnsäure bei. Nur 20 % der Bakterienarten im Darm bauen Harnsäure aktiv ab. Ein Mangel an diesen Darmbakterien führt zu erhöhten Harnsäure-Konzentrationen in Stuhl und Blut.47,48,52Antibiotika stören die Darmflora, verschärfen die Hyperurikämie und erhöhen das Gichtrisiko.49,50,51
Fazit: Ballaststoffreiches Essen und Probiotika stärken die Darmflora. Eine intakte Mikrobiota unterstützt den Abbau von Harnsäure und begünstigt einen ausgewogenen Harnsäurespiegel im Körper.52
3 Purinstoffwechselstörungen als Krankheitsursache
Störungen im Purinstoffwechsel führen zu sehr unterschiedlichen Krankheitsbildern. Einige betreffen Millionen Menschen weltweit – etwa Gicht oder Harnsäuresteine. Andere entstehen wegen genetischer Defekte und treten aussergewöhnlich selten auf.
Gicht
Gicht entsteht infolge einer Hyperurikämie – eines erhöhten Harnsäurespiegels im Blut. Überschreitet die Harnsäure ihre Löslichkeitsgrenze, kristallisiert sie aus. Die Kristalle treten hauptsächlich in Gelenken oder Nierengewebe auf und verursachen schmerzhafte Entzündungen.17,19,22
Statistisch gesehen erleiden weniger als 36 % der Personen mit Hyperurikämie einen akuten Gichtanfall. Unbehandelt treten die Anfälle zunehmend häufiger auf und gehen oft in eine chronische Gicht über.18,19,20
Neuere Forschungen betonen die multifaktorielle Entstehung dieser Zivilisationskrankheit. Entscheidende Einflussgrössen umfassen:19,20,39,45,59
- Höhe und Art der Purinzufuhr
- Fructosestoffwechsel
- Harnsäurespiegel
- Ausscheidung von Harnsäure über die Nieren
- Urinmilieu (niedriger pH fördert Kristallisation)
Meist ist Gicht mit dem metabolischen Syndrom verbunden. Das heisst Fettleibigkeit, Bluthochdruck, abnorme Blutfette und Störungen des Zuckerstoffwechsels. Häufig treten zusätzliche Komorbiditäten auf. Das entspricht vielfach chronischen Lebererkrankungen, Diabetes, Arthrose und Augenkrankheiten.20
Fructose erhöht unabhängig von der Purinaufnahme den Harnsäurespiegel. Bei isolierter Fructose aus Sirup oder Limonade wirkt der Prozess besonders stark. Der moderate Verzehr natürlicher Fructose aus Obst verursacht hingegen meist keine klinisch relevante Hyperurikämie.45
Alkohol hemmt die renale Ausscheidung und steigert gleichzeitig die Produktion von Harnsäure. Spirituosen und Bier gelten daher als wichtige Risikofaktoren für Gicht.57,58,59
Primäre und sekundäre Gicht
Genetisch oder metabolisch bedingte Hyperurikämie führt zu primärer Gicht. Wenn eine oder mehrere Erkrankungen Gicht verursachen, ist von sekundärer Gicht die Rede. Zur sogenannten Pseudogicht lesen Sie bitte unsere Ausführungen über Gicht, Pseudogicht und Osteoporose.
Harnsäuresteine
Harnsäuresteine blockieren die Harnwege und verursachen schmerzhafte Koliken. Sie entstehen, weil der Körper zu viel Harnsäure produziert oder die Nieren sie nicht ausreichend ausscheiden. Ein dauerhaft saurer Urin gilt als entscheidender Risikofaktor.17,43
Harnsäuresteine, Harnsteine oder Nierensteine?
Bei Purinstoffwechselstörungen entstehen Harnsäuresteine grösstenteils in der Niere. Deshalb spricht die Fachliteratur vorwiegend von Nierensteinen (engl. kidney stones). Sie können aber im Harnleiter steckenbleiben oder bis in die Harnblase gelangen. Dann passt auch die allgemeinere Bezeichnung Harnsteine (urinary stones). Zur Betonung der chemischen Zusammensetzung dient der Begriff Harnsäuresteine (uric acid stones) – eine Bezeichnung, die sie von anderen Steinarten wie Calciumsteinen abgrenzt.
Ein höherer Urin-pH erhöht die Löslichkeit von Harnsäure deutlich. Der angestrebte pH-Wert liegt zwischen 6,2 und 6,8. Nützliche Massnahmen:
- Eine purinreduzierte Ernährung senkt die Harnsäureproduktion deutlich.17
- Weniger Protein verhindert zusätzlich eine Übersäuerung des Urins.60,61
- Viel Gemüse und pflanzenbasierte Kost wirken alkalisierend.60,61,67,68,69
- Ausreichend, aber nicht übermässig viel Wasser trinken.
Personen mit genügender Urinmenge entwickeln praktisch keine Steinbildung. Die Konzentration der Stoffe ist zu gering. Die Harndichte (spezifisches Gewicht) beschreibt, wie konzentriert der Urin ist. Sie spiegelt den Flüssigkeitshaushalt genauer wider als die Trinkmenge. Werte zwischen 1,010 und 1,030 g/mL gelten als ideal und zeigen eine gute Nierenfunktion sowie eine ausgeglichene Wasserbilanz. Ab einer Dichte von 1,030 gilt der Urin als stark konzentriert. Das passiert meist infolge geringer Flüssigkeitszufuhr, übermässiger Eiweiss- und Salzaufnahme oder bestimmter Krankheiten.46
Wer überwiegend Gemüse und Früchte isst, nimmt allein über die Nahrung grosse Flüssigkeitsmengen auf. Rechnen Sie mit 1,6 bis 1,8 Liter Wasser pro 2 Kilogramm an naturbelassenen pflanzlichen Lebensmitteln. Damit ist der Wasserbedarf meist gedeckt, sofern die Nieren täglich 1 bis 1,5 Liter Urin ausscheiden. Zusätzliche Trinkmengen dürfen entfallen. Die Trinkmengen-Frage erläutern wir im Abschnitt Wie Leber und Nieren gesund halten. Entscheidend bleibt die Ausscheidungsmenge: Ungetrübter Urin deutet bei gesunden Menschen auf eine gute Flüssigkeitsversorgung und geringe Urinkonzentration hin.12,17
Gicht und Nierensteine – was Ernährung bewirkt
Zu viel Harnsäure im Blut begünstigt Gicht. Zu viel Harnsäure im Urin fördert harnsäurehaltige Nierensteine. Beides hängt zusammen: Wer an Gicht erkrankt, trägt ein doppeltes Risiko für Nierensteine.64,65
Purinreiche Ernährung, viel Fruchtzucker und Alkohol kurbeln die Harnsäureproduktion an.36,54 Gleichzeitig hemmen Übergewicht und Insulinresistenz die Ausscheidung. Fruchtzucker und Alkohol schädigen auf beiden Wegen.57,58,59
Alle diese Faktoren führen zudem häufig zu einem niedrigen Urin-pH. In saurem Urin kristallisiert Harnsäure leichter aus, was die Bildung von Nierensteinen begünstigt.
Die westliche Ernährungsweise hat negative Auswirkungen auf alle oben genannten Punkte (genetische Ursachen ausgeschlossen).36,54,57,58,59,63,66,69
Pflanzenbasierte, purinbewusste Ernährung wirkt auf mehreren Ebenen positiv – die Harnsäureproduktion sinkt, der Urin-pH steigt, die Nieren arbeiten unbelastet.63,66 Tierische Produkte, Alkohol und Fruchtzucker hingegen sorgen für Stoffwechselstress an diesen Orten.
Fazit: Wer Gicht oder Nierensteinen entgegenwirken will, wählt naturnahe und purinbewusste Pflanzenkost. Sie ist reich an Ballaststoffen, Vitaminen, sekundären Pflanzenstoffen und Mineralstoffen - was harnsäuresenkende sowie alkalisierende Effekte auslöst.68,69,70,72
Willkommene Orientierungshilfen bieten dabei der PRAL-Index oder der NEAP-Score. Weshalb Sie selbst diese Werkzeuge kritisch handhaben sollten, erfahren Sie in der detaillierten Textfassung.60,67,70,71,72,73,74
Alterungsprozesse
In moderaten Konzentrationen wirkt Harnsäure antioxidativ.53,54 Chronisch erhöhte Harnsäurewerte hingegen erzeugen oxidativen Stress und aktivieren Entzündungsreaktionen. Diese Veränderungen stehen in direktem Zusammenhang mit altersbedingten Erkrankungen wie Bluthochdruck und Herz-Kreislauf-Leiden.52,55
Optimum statt Minimum oder Maximum gilt als wichtiger Schlüssel zur Gesundheit. Ob zu niedrige oder zu hohe Werte – beide Extreme gehen gleichermassen mit gesundheitlichen Risiken einher.55
Seltene genetische Defekte
Einige Enzymdefekte im Purinstoffwechsel verursachen schwerwiegende Störungen.
- ADSL-Defizienz: Epilepsie, geistige Behinderung, Bewegungsstörungen – oft tödlich im Kindesalter.23
- Lesch-Nyhan-Syndrom: Extreme Harnsäureproduktion mit Gicht, Nierensteinen und schwerem Selbstverletzungsverhalten.24
- Adenosindeaminase-Mangel: Schwere Immundefizienz, bisweilen zusätzlich mit Hörverlust und neurologischen Schäden – unbehandelt tödlich.25
Diese Krankheitsbilder verdeutlichen: Enzyme im Purinstoffwechsel beeinflussen Wachstum, Nervensystem und Immunabwehr.
Purine in der Therapie
Allopurinol hemmt das Enzym Xanthinoxidase und senkt so die Bildung von Harnsäure. Es lindert Gicht und Harnsäuresteine.17
Azathioprin und 6-Mercaptopurin zählen zu den klassischen Purinanaloga. Sie blockieren die DNA-Synthese und bremsen die Vermehrung von Immunzellen. Ärztinnen und Ärzte setzen sie in der Transplantationsmedizin und bei Autoimmunerkrankungen ein.26,27
Eingriffe in den Purinstoffwechsel gewinnen zunehmend an therapeutischer Bedeutung – auch in der Onkologie.28,29
4 Purinarme Ernährung neu definiert
Eine dauerhaft purin- und eiweissreiche Ernährung belastet Leber und Nieren. Bleibt der Harnsäuregehalt dauerhaft erhöht, steigt das Risiko für Gicht und Harnsäuresteine. Eine purin- und proteinreduzierte Ernährung entlastet diese Organe und trägt zu deren Gesundheit bei.
Gibt es Richtwerte für die tägliche Purinzufuhr? Der Grossteil der Purine im Körper stammt aus eigener Synthese oder aus körperinternem Recycling. Daher ist die Definition einer Untergrenze bzw. einer minimalen Tagesaufnahme nicht sinnvoll. Ebenso wenig existieren offiziell standardisierte Obergrenzen. Der Einfluss von diversen Faktoren auf Purinstoffwechsel und Harnsäurespiegel ist zu komplex20,52,59 für eindeutige Fixpunkte.
Zur groben Orientierung dienen Ernährungsratgeber für Personen mit Nierenkrankheiten und Gicht. Diese arbeiten mit Leitlinien zur Risikoreduktion.
- Die Website der Renal Associates klassifiziert folgendermassen: Eine omnivore Standardernährung liefert täglich 600-1000 mg Purine. Bei einer streng purinarmen Ernährung liegen die täglichen Purinmengen bei rund 100-150 mg pro Tag.
- Klinische Ernährungsempfehlungen zur Prävention und Therapie der Hyperurikämie definieren häufig eine ungefähre Höchstgrenze von 400 mg an Purinen pro Tag. Idealerweise stammen diese nicht aus Fleisch, Fisch und Meeresfrüchten, sondern aus pflanzlichen Lebensmitteln.32
Purinarm oder purinbewusst? Moderne Ernährungsleitlinien kritisieren das Konzept der "purinarmen Diät" als veraltet. Sie unterstreichen: Die grössten Vorteile entstehen aus purinbewusster, ausgewogener und pflanzenbasierter Kost.57,58,59 Eine sinnvolle Ernährungsumstellung erfordert mehr, als lediglich purinreiche Lebensmittel zu meiden.
Tierische Purinquellen enthalten oft ungünstige Begleitstoffe – das erhöht tendenziell den Harnsäurespiegel. Pflanzliche Lebensmittel wirken tendenziell protektiv. Deshalb gilt es, den Verzicht auf tierisches Purin zu priorisieren.62
Puringehalt in Lebensmitteln
In der Regel liefern proteinhaltige Lebensmittel viele Purine – primär Fleisch, Fisch und Hülsenfrüchte. Milchprodukte enthalten viel Eiweiss, kaum Purine. Gemüse, Obst und Knollen mit geringem Eiweissgehalt beinhalten meist wenig Purine.
Ausnahmen bilden Spinat, Blumenkohl und Brokkoli. Obwohl ihr Eiweissgehalt gering ist, enthalten sie vergleichsweise viele Purine. Ihre Begleitstoffe mildern aber die Harnsäurewirkung deutlich.
Hülsenfrüchte bringen mittlere Purinmengen mit, gelten jedoch aufgrund ihrer restlichen Inhaltsstoffe als gut verträglich. Vorsicht: In vielen Purin-Tabellen stehen nur Werte für getrocknete, nicht für gekochte Hülsenfrüchte. Beim Kochen geht ein Teil der Purine ins Kochwasser über – der Puringehalt pro 100 g sinkt deutlich.
Warum pflanzliche Purinquellen gesünder wirken
Der Körper verwertet Purin aus tierischen und pflanzlichen Lebensmitteln unterschiedlich.
Epidemiologische Daten bestätigen: Der Verzehr von Fleisch und Fisch erhöht das Risiko für Hyperurikämie und Gicht signifikant. Ein moderater Verzehr von purinhaltigem Gemüse führt hingegen nicht zu einem erhöhten Risiko.38 Während einer Cross-over-Untersuchung von 2012 löste eine purinreiche pflanzliche Ernährung nur selten einen akuten Gichtanfall aus. Tierische Lebensmittel mit viel Purin erhöhten bei den ProbandInnen das Risiko für Gichtschübe um das Fünffache.39
Pflanzenkost bringt zahlreiche Vorteile:
- Ballaststoffe in pflanzlichen Lebensmitteln verlangsamen die Aufnahme von Purinen im Darm.40
- Sekundäre Pflanzenstoffe wie Flavonoide und Anthocyane hemmen das Enzym Xanthinoxidase und reduzieren die Umwandlung von Purin zu Harnsäure.40,41
- Vitamin C steigert die Ausscheidung von Harnsäure über den Urin.40
Folgende Unterschiede erklären, warum der Körper tierische und pflanzliche Purinquellen ungleich verarbeitet:
- Begleitstoffe aus pflanzlichen Lebensmitteln hemmen die Harnsäurebildung oder fördern die Ausscheidung.40,41
- Der Körper verstoffwechselt Purine aus Fleisch und Fisch besonders rasch. Dies ist ein Nachteil: Pro aufgenommene Purinmenge bedeutet das mehr Harnsäure und ein höheres Risiko für Gicht.32,75
- Viele tierische Lebensmittel, v.a. Muskelfleisch und Fisch, enthalten neben den Purinen Adenin und Guanin auch nennenswerte Mengen des Abbauprodukts Hypoxanthin. Adenin und Guanin baut der Körper über mehrere Schritte zu Harnsäure ab. Hypoxanthin hingegen wandelt er mithilfe des Enzyms Xanthinoxidase schneller in Harnsäure um. Deshalb wirkt es biochemisch "direkter".32,75
Verwendung von Purintabellen
Wer purinbewusst isst, nutzt Purintabellen als unverzichtbares Planungsinstrument. Allerdings erlauben viele Angaben keinen direkten Vergleich. In der Praxis existieren unterschiedliche Methoden zur Darstellung von Purinwerten in Lebensmitteln. Sie dienen der Orientierung, nicht der exakten Vorhersage der Harnsäurekonzentration. Zudem weichen sie häufig in Genauigkeit und Aussagekraft voneinander ab. Unterschiedliche Messmethoden, Zubereitung, Wassergehalt und Portionsgrössen führen zu fluktuierenden Werten.
Tabellen geben Puringehalte vielfach pro 100 g an. Jedoch variiert der Kalorien- und Wassergehalt pflanzlicher Lebensmittel bedeutend. Deshalb finden Sie in unseren Tabellen neben dem Wert pro 100 g stets Angaben pro Kilokalorie. Diese Angabe lässt einen besseren Vergleich zwischen Lebensmitteln mit ähnlichem Kaloriengehalt zu. Im Alltag erleichtert der Purinwert pro 100 g die Berechnung der täglichen Aufnahme.
Gleiche Lebensmittel mit unterschiedlichem Puringehalt? Auch für ein und dasselbe Lebensmittel weichen die Angaben in Purin-Tabellen häufig voneinander ab – wegen biologischer und analytischer Faktoren. Das betrifft z.B. Messverfahren, verschiedene Pflanzenteile, Reifegrad oder Zubereitungsart. Die Zahlen variieren teils um mehr als das Doppelte. Details zu den Berechnungsmethoden können Sie im ausführlichen Purin-Beitrag nachlesen.
Wie stark die Werte schwanken, erkennen Sie am Beispiel Brokkoli. Je nach Datenquelle liegen die Angaben zwischen etwa 41 und 70 mg pro 100 g (Tabelle 1).
Tabelle 1: Beispiel Brokkoli – Unterschiede nach Quelle
Tierische Lebensmittel
Fisch, Geflügel, Rind- und Schweinefleisch dominieren mit viel Purin (Tabellen 2a–2c). Im Gegensatz dazu enthalten Milchprodukte wie Joghurt und Käse wenig Purin, ebenso Hühnereier.
Tabelle 2a: Purin in Fleisch
Tabelle 2b: Purin in Fischen
Tabelle 2c: Purin in Milchprodukten und Eiern
Daten zur Tabelle: Purine (mg/100g) aus USDA and ODS-NIH Database for the Purine Content of Foods Release 2.0 (2025), Werte mit ** stammen aus Wolfram&Colling 1987, berechnet aus Harnsäure (Mittelwert) durch 1,1 bzw. 1,25 (stöchiometrisches Verhältnis).
Kalorien (kcal/100g) aus USDA Food Data, ausser Werte mit * = FDDB: Kalorientabelle und Ernährungstagebuch.
Pflanzliche Lebensmittel
Pflanzliche Lebensmittel enthalten in der Regel wenig Purin. Ausnahmen wie Algen, Petersilie und gewisse Pilze sind purinreicher – ebenso Spinat, Gemüsepaprika, Blumenkohl, Kürbis und Brokkoli (Tabelle 3).
Tabelle 3: Pflanzliche Lebensmittel mit viel Purin
Hülsenfrüchte und Getreide(-produkte)
Die meisten Tabellen geben ihre Purinwerte in getrockneter Form an. Beim Kochen übernehmen diese Lebensmittel Wasser und quellen auf – der Puringehalt pro 100 g sinkt deutlich (Tabelle 4). Besonders viel Purin enthalten Weizenkeime. Im Allgemeinen gilt das für alle keimenden Lebensmittel.30
Tabelle 4: Purin in Hülsenfrüchten und Getreide(-produkten)
Gemüse und Kräuter
Der Puringehalt der meisten Gemüsesorten liegt unter 50 mg pro 100 g. Besonders wenig Purin enthalten Zucchini, Gurken, Tomaten, Salate, Süsskartoffeln, Mais und Karotten (Tabellen 5a–5b).
Tabelle 5a: Purin in Gemüse und Kräutern
Tabelle 5b: Purin in Gemüse und Kräutern
Pilze und Algen
Der Puringehalt von Pilzen und Algen variiert je nach Sorte und Zubereitungsart stark (Tabelle 6). Eine purinarme Ernährung schliesst Austern-Seitlinge besser aus. Rohe Champignons, Enoki oder Shiitake wirken vorteilhafter.
Tabelle 6: Purin in Pilzen und Algen
Samen
Samen enthalten, bezogen auf den Energiegehalt, wenig Purin – meist unter 0,3 mg pro kcal (Tabelle 7). Daher verursachen Samen im Purinstoffwechsel keine Probleme und passen gut in eine purinbewusste Ernährung.
Tabelle 7: Purin in Samen
Nüsse
Nüsse beinhalten wenig Purin. Mit 0,03–0,08 mg/kcal tragen sie kaum zur Harnsäurebelastung bei. Bei erhöhtem Harnsäurespiegel oder Gicht bieten sie eine gut verträgliche Eiweiss- und Fettquelle (Tabelle 8).
Tabelle 8: Purin in Nüssen
Früchte
Frische Früchte liefern wenig Purin, meist unter 40 mg pro 100 g.31 Trockenobst ist purinreicher, da alle Nährstoffe konzentrierter vorliegen. Fruchtsäfte weisen zwar niedrige Puringehalte auf – über die Fructose erhöhen sie trotzdem das Gichtrisiko.36 Menschen mit Gicht meiden Fruchtsäfte mit Vorteil.
Verzichten Sie nie auf frische Früchte – insbesondere auf reifes, nährstoffreiches Obst. Ein höherer Verzehr von gesunder, pflanzenbasierter Kost geht mit einem wesentlich geringeren Gichtrisiko einher.34,35,37
5 Gesund und purinbewusst essen: die Grundlagen
Unsere Bilanz: Purinbewusste Menschen verzichten auf Fleisch und Fisch. Eier, Milch und Milchprodukte enthalten wenig Purin – dennoch spricht ethisch und gesundheitlich vieles gegen ihren Verzehr. Fruchtsäfte und Lebensmittel mit freier Fructose – etwa Agavensirup, Maissirup oder gesüsste Getränke – gehören vom Speiseplan gestrichen.
Idealerweise bleibt Alkohol ganz weg – einschliesslich hefelastiger, alkoholfreier Biere. Ethanol behindert die renale Harnsäureausscheidung und steigert den Laktatspiegel im Blut. Beides trägt zur Erhöhung der Harnsäurewerte bei. Bier enthält je nach Gärung und Hefefilterung 10-150 mg Purin pro 100 g. Alkoholfreie, hefereiche Sorten enthalten teils mehr Purin als gefilterte alkoholische Biere.21,33
Der Puringehalt pflanzlicher Lebensmittel liegt meist niedrig. Purinreiche pflanzliche Quellen verträgt der Körper zudem besser und sie liefern gesundheitsfördernde sekundäre Pflanzenstoffe. Beachten Sie vor allem bei Nierenkrankheiten und metabolischem Syndrom: Purinreiche Quellen nicht kombinieren – etwa Spinat mit gewissen Pilzen oder Blumenkohl mit Petersilie.
Bevorzugen Sie eine ballaststoffreiche Ernährung und konsumieren Sie gelegentlich fermentierte Lebensmittel mit Probiotika. Eine stabile Darmflora fördert den Abbau von Harnsäure im Darm.
Vegane Sojaprodukte enthalten wenig Purin. Normale Sojamilch (ungesüsst) weist rund 19,34 mg/100g auf.44 Tofu enthält in roher Form etwa 31 mg pro 100 g und angebraten 54 mg – beide Male klassifiziert als purinarmes Lebensmittel.30
Vegane Rezepte mit wenig Purin
Pflanzliche Lebensmittel verbinden automatisch Geschmack mit Purinarmut, Nährstoffdichte und Nachhaltigkeit. Eine Auswahl an Rezepten mit tiefer Purinlast finden Sie in unserer Rezept-Datenbank.
Unser Tipp: Bei strikt purinarmer Kost besser Spinat, Brokkoli, Kürbis, Gemüsepaprika, Blumenkohl, Pilze und Algen meiden.
Das Erb-Müesli liefert zum Frühstück eine nährstoffreiche Basis mit geringem Puringehalt pro Kalorie und wertvollen Omega-3-Fettsäuren.
Purinarmes aus der Salatküche passt gut als Hauptgericht oder Beilage:
- Papaya a la Mexicana mit Avocados
- Gesunder, ölfreier Rote-Bete-Salat mit Apfel
- Roh-veganer, ölfreier Zucchinisalat mit Tomate und Basilikum
- Orangen-Fenchelsalat mit Mandeln
- Zitrussalat mit Sumak-Dressing, Berberitzen und Pistazien
- Chicorée-Feigensalat mit Bleichsellerie und Avocados
- Birnen-Rucola-Salat mit Quinoa und Balsamico-Dijon-Dressing
- Griechischer Grünkohl-Salat mit Kichererbsen und Oliven
- Gesunder süsslich-scharfer Grünkohlsalat mit Roter Bete
- Roher Zuckerschotensalat mit Walnüssen und Datteln
Kombinieren Sie Ihre Lieblingssalate mit frischen Früchten und Gemüse. Würzen Sie diese mit einem ölfreien Salatdressing oder einer Vinaigrette.
Probieren Sie eines der folgenden Rezepte für eine schmackhafte, herzhafte und purinbewusste Hauptspeise:
- Gefüllte Zucchini mit Quinoa-Tomaten-Mischung
- Quinoa-Bowl mit Kichererbsen und Mais
- Rosenkohl mit Maronen und Fenchelsamen
- Ackerbohneneintopf mit Dillsauce und gemahlenem Kurkuma
- Feurige roh-vegane Tomatensuppe mit Avocado und Basilikum
- Kräftige Rote-Bete-Suppe mit Fenchel- und Koriandersamen
- Bohneneintopf mit Mangold und Tomaten
- Apfel-Linsen-Dal mit Kurkuma und Kreuzkümmel
- Stuffed Tomatoes mit Hirse, getrockneten Tomaten und Oliven
- Gebackener Wirsing mit Dinkelkörnern, Karotten und Tomaten
- Fruchtige Rote-Bete-Fenchel-Cremesuppe mit Apfel und Kurkuma
Frische Früchte passen ideal zu kleinen Zwischenmahlzeiten oder als süsser Abschluss. Etwa das Erb-Resveratrol-Dessert mit dunklen Trauben. Desserts wie rohe Schokoladencrème oder roh-veganer Blaubeerkuchen überzeugen mit geringem Purinanteil.
Literaturverzeichnis - 75 Quellen (Link zur Evidenz)
| 1. | * Narratives Review Purines are ubiquitous biomolecules that sustain life. Purines are incorporated into DNA and RNA, found as the energy currency of cells (ATP and GTP), used as sig naling molecules (ATP, cAMP, and cGMP), and inte grated into coenzymes (FAD, NADþ, NADPþ, and coenzyme A). These purines are generated by either or both of the two pathways: de novo purine biosynthesis (DNPB) or purine salvage. DOI: 10.1080/10409238.2020.1832438 Study: weak evidence | Pareek V, Pedley AM, Benkovic SJ. Human de novo purine biosynthesis. Crit Rev Biochem Mol Biol. 2021;56(1):1-16. |
| 2. | * experimentelle Studie In mammals, the two primary routes for the synthesis of the adenine nucleotides ATP, ADP and AMP, and, as a consequence, the major bioactive metabolite adenosine, are the de novo purine biosynthesis (DNPB) pathway, and the purine salvage pathway (PSP). Of the two, the PSP dominates in both the mammalian brain and heart. This is because the PSP utilizes the breakdown products of ATP, occasioned by the high energy demands of these organs, to rapidly regenerate adenine nucleotides. This resynthesis route, while efficient and energetically favourable, leaves these organs vulnerable to loss of salvageable metabolites, with the potential for protracted depletion of the means to synthesize ATP, and the ability to deploy neuro- and cardioprotective adenosine. DOI: 10.1016/j.neuropharm.2022.109370 Study: weak evidence | Gessner P, Lum J, Frenguelli BG. The mammalian purine salvage pathway as an exploitable route for cerebral bioenergetic support after brain injury. Neuropharmacology. 2023;224:109370. |
| 3. | ● The dominance of nucleic acids and molecular biology in contemporary biochemistry tends to obscure the fact that biological and chemical knowledge of the purines substantially predated their discovery as nucleic acid constituents and can be traced back over 200 years. The first of these compounds to be isolated was uric acid (1) obtained by Scheele and Bergman in 1776 from bird excreta, human urine and urinary calculi. Undoubtedly this early discovery was greatly facilitated by the relatively low solubility of uric acid and hence its tendency to crystallize easily from biological fluids and extracts. Interest in the chemistry of urinary calculi also led Marcet, some forty years later, to the discovery of xanthine (2). Guanine (3) was isolated by Magnus in 1844 from guano, hence the name, and this discovery was followed in 1850 by Scherer’s isolation of hypoxanthine (4) from beef spleen. The last of the commonly occurring purines to be discovered was adenine (5) obtained by Kossel in 1885-6 from beef pancreas. The dominance of nucleic acids and molecular biology in contemporary biochemistry tends to obscure the fact that biological and chemical knowledge of the purines substantially predated their discovery as nucleic acid constituents and can be traced back over 200 years. The first of these compounds to be isolated was uric acid (1) obtained by Scheele and Bergman in 1776 from bird excreta, human urine and urinary calculi. Undoubtedly this early discovery was greatly facilitated by the relatively low solubility of uric acid and hence its tendency to crystallize easily from biological fluids and extracts. Interest in the chemistry of urinary calculi also led Marcet, some forty years later, to the discovery of xanthine (2). Guanine (3) was isolated by Magnus in 1844 from guano, hence the name, and this discovery was followed in 1850 by Scherer’s isolation of hypoxanthine (4) from beef spleen. The last of the commonly occurring purines to be discovered was adenine (5) obtained by Kossel in 1885-6 from beef pancreas. DOI: 10.1007/978-94-011-4906-8_6 Book: strong evidence | Brown EG. Purines. Ring Nitrogen and Key Biomolecules. 2012:128-166. |
| 4. | ●
Book: moderate evidence | Martz E. Timeline of Protein Chemistry. 2002. Zusammenfassung aus dem Buch Tanford Ch, Rynolds J. Nature's Robots - A History of Proteins. Oxford University Press, 2001. |
| 5. | ● 2.1 Discovery of Purines and Pyrimidines The history of purines and pyrimidines began in 1776 when the Swedish pharmacist Carl Wilhelm Scheele isolated uric acid from bladder stones (Scheele 1776). Almost seven decades later, in 1844, guanine was isolated by Unger from the faeces of Peruvian guano sea birds (Unger 1846). At the end of the nineteenth century, several principal purines (adenine, xanthine and hypoxantine) and pyrimidines (thymine, cytosine and uracil) were discovered by Ludwig Karl Martin Leonhard Albrecht Kossel (1853–1927; see Jones 1953; Bendich 1955; Persson 2012; the original Kossel report appeared in Chem. Ber., 1885, 18, 79). Interestingly, already at that stage it was believed that these substances constitute the main part of cell nuclei; Kossel followed experimental protocols of Friedrich Miescher (1844–1895), who was the first to isolate the nuclear material rich in phosphorus that was called ‘nuclein’ (Miescher 1874; Hoppe-Seyler 1871). In the same period the great Emil Fischer started to investigate the structure of caffeine and related compounds (Fischer 1881). He solved the structures and confirmed them by synthesis. It was also Emil Fischer who, based on his structural studies, introduced the term ‘purines’ (purum uricum) (Fischer 1907); this was one of the reasons for his Nobel Prize in 1902. The term ‘pyrimidines’ was introduced by (Pinner 1885). An arduous task of determining the sugar part of nucleosides (and nucleotides) followed and was finally solved by Phoebus Aaron Levene (Levene and Jacobs 1908; Levene and Tipson 1931). In 1927, Gustav Embden and Margarete Zimmermann described adenosine monophosphate in skeletal muscle (Embden and Zimmermann 1927). Adenosine 50 -triphosphate (ATP) was discovered in 1929, independently by Karl Lohmann in Germany and by Cyrus Hartwell Fiske and Yellagaprada SubbaRow in the USA (Fiske and SubbaRow 1929; Lohmann 1929). Lohman (1898–1978) was in those days working as the assistant of Otto Meyrhoff in Berlin; Fiske (1890–1978) was an associate professor in Harvard Medical School in Boston, and SubbaRow (1896–1948) was Fiske’s PhD student (Fig. 2.1). Lohman’s publication appeared several months earlier (in August 1929) than the paper by Fiske and SubbaRow (which was published in October 1929), and yet the latter had obtained the first evidence for ATP probably as early as 1926. It all came to a climax in August 1929, during the thirteenth Physiological Congress in Boston when Lohman and Fiske discussed the priority matters. Whether Fiske briefed Otto Meyerhof, who was Lohmann’s director, about his discovery (and then Meyerhof pushed Lohman’s publication) or not, remains a matter of doubt (the dramatic history of ATP discovery is described in detail in Maruyama 1991). In the following decade, the role of ATP in cell energetics was firmly established and the concept of the ‘high-energy phosphate bond’ was introduced by Fritz Lipman (Lipman 1941). DOI: 10.1007/978-3-642-28863-0_2 Book: strong evidence | Burnstock G, Verkhratsky A. Early History of Purinergic Signaling. In: Purinergic Signalling and the Nervous System. Berlin/Heidelberg: Springer;2012:7-66. |
| 6. | * Historische Betrachtung und Geschichte zu Protein. Hatte Hofmeister die amidartige Verknüpfung der Aminosäuren in den Proteinen postuliert, so erbrachte Emil Fischer (1852-1919)82 dafür den exakten chemischen Beweis. Emil Fischer erhielt den Nobelpreis für Chemie 1902 für „seine synthetischen Arbeiten auf dem Gebiet der Zucker und der Purine“. DOI: - | Schwenke KD. Vom Eyweiss zum Protein: Zur Geschichte eines Begriffes. Mitteilungen Gesellschaft Deutscher Chemiker. Frankfurt/Main;2007;19:25-49. |
| 7. | ● Proteins are made up of 20 amino acids. Each amino acid has an α-carboxyl group, a primary α-amino group, and a side chain called the R group (see Image. Amino Acid Generic Structure). Unlike other amino acids, proline has a secondary amino group. The side chain varies from 1 amino acid to the other. Nutritionally, amino acids are divided into 3 groups—essential, nonessential, and semi-essential. Semi-essential amino acids are synthesized by the body but are designated essential during periods of stress. Nine amino acids, including histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine, are classified as essential amino acids because they cannot be synthesized by human or other mammalian cells. Therefore, these amino acids must be supplied from an exogenous diet. Besides 20 amino acids that participate in protein synthesis, recently, 2 more new amino acids have been described—selenocysteine and pyrrolysine. Selenocysteine occurs at the active site of several enzymes, including thioredoxin reductase and glutathione peroxidase. Pyrrolysine is not present in humans but is used in the biosynthesis of proteins in some methanogenic species, such as archaea and bacteria. Website | Lopez MJ, Mohiuddin SS. Biochemistry, essential amino acids. In: StatPearls. National Library of Medicine. 2024. |
| 8. | * Narratives Review For at least 60 years, it has been the convention to divide amino acids into two categories: indispensable (or essential) and dispensable (or nonessential). This categorization provides a convenient, and generally useful, way of viewing amino acid nutrition. However, despite the longevity of the convention, as more information has become available, the distinctions between dispensable and indispensable amino acids, at least at the metabolic level, have become increasingly blurred. According to this restricted metabolic definition of essentiality, threonine and lysine (and perhaps tryptophan) are the only truly essential amino acids. the original nutritional definition of an indispensable amino acid was, “One which cannot be synthesized by the animal organism out of materials ordinarily available to the cells at a speed commensurate with the demands for normal growth.“ This is because some indispensable amino acids can be synthesized from precursors that are structurally very similar. For example, methionine can be synthesized both by transamination of its keto acid analogue and by remethylation of homocysteine. In this sense, then, the mammal is capable of synthesizing leucine, isoleucine, valine, phenylalanine and methionine. However, this is not new synthesis, because the branched-chain keto acids and homocysteine were originally derived from branched-chain amino acids and methionine, respectively. According to this restricted metabolic definition of essentiality, threonine and lysine (and perhaps tryptophan) are the only truly essential amino acids.
DOI: 10.1093/jn/130.7.1835S Study: weak evidence | Reeds PJ. Dispensable and indispensable amino acids for humans. J Nutr. 2000;130(7):1835S–1840S. |
| 9. | ● Nucleic Acid Digestion The nucleic acids DNA and RNA are found in most of the foods you eat. Two types of pancreatic nuclease are responsible for their digestion: deoxyribonuclease, which digests DNA, and ribonuclease, which digests RNA. The nucleotides produced by this digestion are further broken down by two intestinal brush border enzymes (nucleosidase and phosphatase) into pentoses, phosphates, and nitrogenous bases, which can be absorbed through the alimentary canal wall. The large food molecules that must be broken down into subunits are summarized in Table 2. Website | LumenLearning: Chemical Digestion and Absorption: A Closer Look. |
| 10. | * Narratives Review Here, we review the current understanding of the intestinal absorption of nucleobases and analogs. This includes recent knowledge about the efflux transport of those compounds across the basolateral membrane when exiting epithelial cells, following brush border uptake, in order to complete the overall absorption process; the facilitative transporters of equilibrative nucleoside transporter 1 (ENT1/SLC29A1) and equilibrative nucleobase transporter 1 (ENBT1/SLC43A3) may be involved in that in many animal species, including human and rat, without any major species differences. DOI: 10.1248/bpb.b20-00342 Study: weak evidence | Yuasa H, Yasujima T, Inoue K. Current Understanding of the Intestinal Absorption of Nucleobases and Analogs. Biol Pharm Bull. 2020;43(9):1293-1300. |
| 11. | ● Kapitel in Fachlexikon (Enzyklopädie-Beitrag) Purine nucleotide degradation starts with nucleoside for mation (adenosine, inosine, and guanosine) through removal of phosphate moieties (Figure 3) mainly catalyzed by the 50 nucleotidase enzymes. Inosine and guanosine, through the action of purine-nucleoside phosphorylase, are transformed into the purine bases hypoxanthine and guanine, respectively. Adenosine is converted into inosine by the enzyme adenosine deaminase. Guanine, on the other hand, is converted to xanthine by guanine deaminase. Finally, the purine bases hypoxanthine and xanthine (oxypurines) are oxidized to urate by the enzyme xanthine oxidoreductase, one of its iso forms being xanthine oxidase, which is a substrate for com monly used urate-lowering gout medications. In general, the activity of these enzymes is regulated by substrate availability. In humans and primates, urate is the final product of purine metabolism, but in most other animals, urate is degraded to allantoin by the enzyme uricase. DOI: 10.1016/B978-0-12-386456-7.04303-3 Book: moderate evidence | Gaffo AL. Crystal Diseases. In: McManus LM, Mitchell RN. Pathobioloogy of Human Disease. 2014:1935-1949. |
| 12. | * Narratives Review At physiologic pH, uric acid is a weak acid with a pKα of 5.8. Uric acid exists majorly as urate, the salt of uric acid. As urate concentration increases in blood, uric acid crystal formation increases. The normal reference interval of uric acid in human blood is 1.5 to 6.0mg/dL in women and 2.5 to 7.0mg/dL in men. The solubility of uric acid in water is low, and in humans, the average concentration of uric acid in blood is close to the solubility limit (6.8mg/dL). When the level of uric acid is higher than 6.8mg/dL, crystals of uric acid form as monosodium urate (MSU). Humans cannot oxidize uric acid to the more soluble compound allantoin due to the lack of uricase enzyme. Normally, most daily uric acid disposal occurs via the kidneys [2]. The production and catabolism of purines are relatively constant between 300 and 400mg per day. The kidneys eliminate approximately two-thirds, while the gastrointestinal tract eliminates one-third of the uric acid load. Almost all uric acid is filtered from glomeruli, while post-glomerular reabsorption and secretion regulate the amount of uric acid excretion. The proximal tubule is the site of uric acid reabsorption and secretion, and approximately 90% is reabsorbed into blood. This is primarily accomplished at the proximal tubular level by transporters that exchange intracellular anions for uric acid. Almost all reabsorption of uric acid occurs at the S1 segment of the proximal tubule. In the S2 segment of the proximal tubule, uric acid is secreted to a greater extent than that which undergoes reabsorption. Post-secretory reabsorption occurs at a more distal site of the proximal tubule, and approximately 10% of the filtered uric acid appears in the urine [1].
The kidneys eliminate approximately two-thirds, while the gastrointestinal tract eliminates one-third of the uric acid load. Almost all uric acid is filtered from glomeruli, while post-glomerular reabsorption and secretion regulate the amount of uric acid excretion. The proximal tubule is the site of uric acid reabsorption and secretion, and approximately 90 % is reabsorbed into blood. Almost all reabsorption of uric acid occurs at the S1 segment of the proximal tubule. In the S2 segment of the proximal tubule, uric acid is secreted to a greater extent than that which undergoes reabsorption. Post-secretory reabsorption occurs at a more distal site of the proximal tubule, and approximately 10 % of the filtered uric acid appears in the urine. DOI: 10.1016/j.ijcard.2015.08.109 Study: weak evidence | Maiuolo J, Oppedisano F et al. Regulation of uric acid metabolism and excretion. Int J Cardiol. 2016;213:8-14. |
| 13. | * Beobachtungsstudie mit 123 anurischen Hämodialysepatienten Serum urate levels (SUAs) are regulated by the balance between production and excretion of uric acid. Urate is excreted via renal and extra-renal pathways, the latter mainly involving the intestinal tract. According to radio-isotope experiments conducted more than half a century ago1,2,3,4,5,6,7,8,9,10,11,12, roughly two-thirds of urate excretion occurs via the renal pathway, and the remaining one-third mainly via the intestine in normal individuals. In the intestine, uric acid is also excreted by several urate transporters including ABCG219,20,21,22. The expression of ABCG2 in the intestine is remarkable compared to other intestinal urate transporters (e.g. SLC2A9, SLC17A4, and ABCC4) (browsed THE HUMAN PROTEIN ATLAS, https://www.proteinatlas.org/, 2022/8/17). Additionally, it has been reported that single nucleotide polymorphisms (SNPs) of ABCG2 have an order of magnitude greater impact on the SUA and gout than do the SNPs of other urate transporters expressed in the intestine in the general population23,24. The impact of ABCG2 SNPs was even greater in chronic kidney disease patients who have lower renal urate excretion than in the general population24,25. Indeed, there are many papers suggesting that ABCG2 is the major exporter in extra-renal urate excretion26,27,28,29,30,31,32. SUA and PoolUA increased significantly with ABCG2 dysfunction, and extra-renal ABCG2 could excrete up to approximately 60% of the daily uric acid turnover in hemodialysis patients. Our findings indicate that the extra-renal urate excretion capacity can expand with renal function decline and highlight that the extra-renal pathway is particularly important in the uric acid homeostasis for patients with renal dysfunction. In other words, extra-renal ABCG2 can excrete up to approximately 60% of the uric acid production in response to decreased renal function. Therefore, ABCG2-mediated urate excretion in the intestine would serve an important role in compensating for the loss of renal urate excretion under conditions of decreased or lost renal function. DOI: 10.1038/s41598-022-26519-x Study: moderate evidence | Ohashi Y, Toyoda M et al. Evaluation of ABCG2-mediated extra-renal urate excretion in hemodialysis patients. Sci Rep. 2023;13(1):93. |
| 14. | ● Kapitel in Buch Book: strong evidence | Matthews DE. Proteins and amino acids. In: Shils ME, Olson JA, Shike M, Ross AC, editors. Modern Nutrition in Health and Disease. 9th ed. Philadelphia: Lippincott, Williams & Wilkins; 1999. |
| 15. | * Prospektive Beobachtungsstudie (Kohortenstudie) DOI: 10.1093/ndt/gfab036 Study: moderate evidence | Wagner S, Merkling T et al. Water intake and progression of chronic kidney disease: the CKD-REIN cohort study. Nephrol Dial Transplant. 2022;37(4):730-739. |
| 16. | * Narratives Review We previously demonstrated that some hyperuricemic patients who were being treated with an XOR inhibitor still had high plasma XOR activities independent of uric acid levels, indicating resistance to an XOR inhibitor (17). Those subjects were being treated for diabetes mellitus and/or had obesity and liver dysfunction. In addition, plasma XOR activities have been reported to be unexpectedly high in some female subjects with uric acid levels of <4.0 mg/dL who had insulin resistance and/or liver dysfunction (18). These findings indicate the possible significance of lowering plasma activity of XOR, not only lowering uric acid level. Rather than reducing uric acid level, inhibiting plasma XOR activity could be a novel therapeutic strategy for cardiovascular and metabolic diseases DOI: 10.1152/ajpendo.00378.2020 Study: weak evidence | Furuhashi M. New insights into purine metabolism in metabolic diseases: role of xanthine oxidoreductase activity. American Journal of Physiology-Endocrinology and Metabolism. 2020;319(5):E827–E834. |
| 17. | * Why most patients with gout present with acidic urine yet only 20% have uric acid stone formation remains unclear. Narratives Review uric acid stones: Hyperuricuria, low urinary output and acidic urine are well known contributing factors. However, the most important factor for uric acid stone formation is persis tently acidic urine.
Uric acid is the end product of purine metabolism in humans. In other mammals uric acid is further broken down into allantoin by the enzyme uricase. Allantoin is 10 to 100 times more soluble compared with uric acid. Humans and Dalmatian dogs are the only known mammals prone to uric acid stone formation. However, the mechanism of stone for mation in the Dalmatian dog is related to an increased frac tional excretion of uric acid
Two factors contrib ute to uric acid solubility: uric acid concentration and solu tion pH. However, the solubility of uric acid in urine is primarily determined by urinary pH. The first pKa of uric acid is at a pH of 5.5, resulting in the loss of 1 proton from uric acid and the formation of anionic urate.17 The second pKa is 10.3, which has no physiological significance in humans. The supersaturation of urine with uric acid occurs whenurinary pHisless than 5.5. In contrast, at a pH of more than 6.5 the majority of uric acid is in the form of anionic urate (fig. 1).
Urinary alkalization with potassium citrate or sodium bicarbonate is a highly effective treatment, resulting in dissolution of existing stones and prevention of recurrence
The primary treatment modal ity is dietary restriction of purine rich foods. If this approach is unsuccessful, allopurinol is the medication of choice. Po tassium citrate is also effective in preventing calcium oxalate crystallization.45 Patients with symp tomatic hyperuricemia or those not responding to dietary modifications should receive allopurinol. Allopurinol is a xanthine oxidase inhibitor that converts hypoxanthine to xanthine and xanthine to uric acid. Xan thine and hypoxanthine are soluble and are excreted by the kidney. Allopurinol also deceases de novo purine synthesis. Inhibition of purine synthesis does not occur in patients with myeloproliferative disorders or hypoxanthine guanine phos phoribosyl transferase deficiency. Therefore, xanthine stones may form during allopurinol therapy in these individu als.77,78 Oxypurinol is a metabolite of allopurinol. High dose allo purinol therapy rarely has been associated with oxypurinol stones or nephropathy.79 In patients with myeloproliferative disorders allopurinol should be given before chemotherapy to reduce the risk of uric acid stones due to cell lysis. DOI: 10.1016/S0022-5347(05)64439-4 Study: weak evidence | Shekarriz B, Stoller ML. Uric acid nephrolithiasis: current concepts and controversies. J Urol. 2002;168(4 Pt 1):1307-1314. |
| 18. | ● Dazu die Unterseiten Fibromyalgie-Syndrom, Morbus Bechterew, Systemischer Lupus erythematodes (SLE), Glossar, Osteoporose, Rheumatoide Arthritis, Psoriasis-Arthritis, Sjögren-Syndrom, Sklerodermie (systemische Sklerose), Juvenile idiopathische Arthritis (JIA), Vaskulitis, Arthrose, Weichteilrheuma, Gicht, Pseudogicht. Website | Rheumaliga ch: Rheuma von A bis Z. |
| 19. | ● Book: moderate evidence | Hettenkofer HJ (Hrsg.). Rheumatologie: Diagnostik – Klinik – Therapie. 5. Aufl. Stuttgart: Georg Thieme Verlag; 2003. |
| 20. | * Narratives Review Gout, the most common inflammatory joint disease worldwide [1], is characterized by the deposition of monosodium urate (MSU) crystals in joints and surrounding tissues, causing acute pain and inflammation. Recognized since ancient times [2] as the “disease of kings” due to its associations with lifestyle factors, gout’s pathogenesis centers on elevated serum uric acid (SUA) levels, or hyperuricemia, which is now recognized as a primary etiological factor for crystal deposition. The importance of managing gout and hyperuricemia extends beyond simply reducing painful joint flares. Hippocrates, who described gout around 400 BC, observed potential associations with broader health issues [6], a notion that research in the 20th and 21st centuries has supported and expanded. Today, gout and hyperuricemia are recognized as systemic metabolic disorders associated with a range of comorbidities, including cardiovascular diseases, chronic kidney disease, metabolic syndrome, and hepatic steatosis. Hyperuricemia does not necessarily lead to gout. It has been reported that only up to 36% of hyperuricemic individuals develop gout attacks. It has, however, been found that only about half of the individuals with SUA concentrations of ≥600 μmol/L (approximately 10 mg/dL) developed clinically evident gout over a 15-year period [23]. It is not completely clear why some hyperuricemic individuals develop gout attacks and others do not. The mechanisms implicated include the overstimulation of cell proliferation and inflammation, the production of genetic variance in chemotactic cytokines, and the internalization of pro-apoptotic and inflammatory factors induced by extracellular uric acid . DOI: 10.3390/jcm13247616 Study: weak evidence | Timsans J, Palomäki A, Kauppi M. Gout and hyperuricemia: a narrative review of their comorbidities and clinical implications. JCM. 2024;13(24):7616. |
| 21. | * Narratives Review Another cause of overproduction of uric acid relates to acceleration of ATP degradation to AMP, a precursor of uric acid (fi gure 1). This overproduction can arise with excessive alcohol or fructose consumption. DOI: 10.1016/S0140-6736(09)60883-7 Study: weak evidence | Richette P, Bardin T. Gout. The Lancet. 2010;375(9711):318-328. |
| 22. | * Narratives Review While hyperuricemia is a clear risk factor for gout, local factors have been hypothesized to play a role in crystal for mation, such as temperature, pH, mechanical stress, cartilage components, and other synovial and serum factors. Interest ingly, several studies suggest that MSU crystals may drive the generation of crystal-specific antibodies that facilitate future MSU crystallization. Using this definition, hyperuricemia occurs at serum urate levels >6.8 mg/dL [4]. Overly acidic urine is also a critical driver of UA stone formation and is an identifiable risk factor in the majority of UA stone formers [53, 54]. Whereas synovial fluid and/or serum pH are maintained within a narrow range, urine pH can varymorewidely. Ataurinary pHofless than5.5,urinary urate exists largely as UA, the undissociated or protonated form. In contrast to ionized urate, UA is more hydrophobic and less soluble. Concentrations of urate that would be under saturated as an ion become supersaturated as UA, allowing crystals to precipitate. Understanding the process of UA stone formation in a patient can guide treatment. In particular, urine alkalinization is an important approach for stone reduction. Increased fluid intake and reduction of urinary urate excretion through urate lowering medications are less important. less than5.5,urinary urate exists largely as UA, the undissociated or protonated form. DOI: 10.1007/s11926-013-0400-9 Study: weak evidence | Martillo MA, Nazzal L, Crittenden DB. The crystallization of monosodium urate. Curr Rheumatol Rep. 2014;16(2):400. |
| 23. | * Narratives Review Adenylosuccinate lyase ADSL) deficiency is a defect of purine metabolism affecting purinosome assembly and reducing metabolite fluxes through purine de novo synthesis and purine nucleotide recycling pathways. Biochemically this defect manifests by the presence in the biologic fluids of two dephosphorylated substrates of ADSL enzyme: succinylaminoimidazole carboxamide riboside (SAICAr) and succinyladenosine (S-Ado). More than 80 individuals with ADSL deficiency have been identified, but incidence of the disease remains unknown. The disorder shows a wide spectrum of symptoms from slowly to rapidly progressing forms. The fatal neonatal form has onset from birth and presents with fatal neonatal encephalopathy with a lack of spontaneous movement, respiratory failure, and intractable seizures resulting in early death within the first weeks of life. Diagnosis is facilitated by demonstration of SAICAr and S-Ado in extracellular fluids such as plasma, cerebrospinal fluid and/or followed by genomic and/or cDNA sequencing and characterization of mutant proteins. Over 50 ADSL mutations have been identified and their effects on protein biogenesis, structural stability and activity as well as on purinosome assembly were characterized. To date there is no specific and effective therapy for ADSL deficiency. DOI: 10.1007/s10545-014-9755-y Study: weak evidence | Jurecka A, Zikanova M et al. Adenylosuccinate lyase deficiency. Journal of Inherited Metabolic Disease. 2014;38(2):231-241. |
| 24. | * Narratives Review Deficiency of hypoxanthine-guanine phosphoribosyltransferase (HPRT) activity is an inborn error of purine metabolism associated with uric acid overproduction and a continuum spectrum of neurological manifestations depending on the degree of the enzymatic deficiency. Several mechanisms can be identified that contribute to uric acid overproduction in HPRT deficiency [27, 28]. a) HPRT catalyses the salvage synthesis of inosine monophosphate (IMP) and guanosine monophosphate (GMP) from the purine bases hypoxanthine and guanine respectively, utilizing 5'-phosphoribosyl-1-pyrophosphate (PRPP) as a co-substrate (Figure 1). The combination of deficient recycling of purine bases with increased synthesis of purine nucleotides explains marked uric acid overproduction in HPRT deficiency. Neurological manifestations include severe action dystonia, choreoathetosis, ballismus, cognitive and attention deficit, and self-injurious behaviour. Compulsive self-injurious behaviour is the most striking feature of Lesch-Nyhan syndrome and is only present in patients with the complete enzyme defect, although some Lesch-Nyhan patients never show auto-destructive behaviour. DOI: 10.1186/1750-1172-2-48 Study: weak evidence | Torres RJ, Puig JG. Hypoxanthine-guanine phosophoribosyltransferase (HPRT) deficiency: Lesch-Nyhan syndrome. Orphanet J Rare Dis. 2007;2:48. |
| 25. | * Narratives Review Adenosine deaminase deficiency (ADA) is a purine salvage pathway deficiency that results in buildup of toxic metabolites causing death in rapidly dividing cells, especially lymphocytes. The most complete form of ADA leads to severe combined immune deficiency (SCID). Adenosine deaminase deficiency (ADA) is a purine salvage pathway defect leading to toxic buildup of the substates adenosine (Ado) and deoxyadenosine (dAdo) and buildup of dAdo nucleotides (dAXP).1 ADA is expressed in almost all cells but has very high activity in lymphocytes because they are rapidly dividing.1,2 ADA deficiency can lead to sensorineural hearing loss, skeletal defects, and neurodevelopmental deficits, but the immunological manifestations are potentially life-threatening.3,4 ADA severe combined immune deficiency (SCID) results from the most complete form of ADA deficiency where there is <1% ADA activity and usually presents near birth.5 Late onset ADA can also be severe with <1% activity and lead to ADA SCID, or it can be less severe with partial activity causing a combined immune deficiency (CID), and either of these may be missed on T cell receptor excision circle (TREC) newborn screening (NBS) which is done in the first days of life.6 Combined immune deficiency from ADA deficiency may present later in life with varying degrees of B cell, T cell, and NK cell dysfunction. To assure that no cases of ADA SCID are missed at least one state, Michigan, now adds ADA enzyme screening to its newborn screening panel. DOI: 10.2147/TCRM.S350762 Study: weak evidence | Secord E, Hartog NL. Review of Treatment for Adenosine Deaminase Deficiency (ADA) Severe Combined Immunodeficiency (SCID). Ther Clin Risk Manag. 2022;18:939-944. |
| 26. | * Systematisches Review Purine analogues such as azathioprine (AZA) and 6‐mercaptopurine (6‐MP) have been used in clinical practice for over five decades. 6‐MP and its prodrug AZA […] are purine antimetabolites that reduce cell proliferation and have immune modulating properties. 6‐MP is metabolised to its active component 6‐thioguanine nucleotide which competitively interferes with nucleic acid metabolism by inhibiting the proliferation of T and B lymphocytes. Moderate certainty evidence suggests that AZA and 6‐MP may be superior to placebo for maintenance of surgically‐induced remission in participants with Crohn’s disease
DOI: 10.1002/14651858.CD010233.pub3 Study: strong evidence | Gjuladin-Hellon T, Iheozor-Ejiofor Z et al. Azathioprine and 6-mercaptopurine for maintenance of surgically-induced remission in Crohn's disease. Cochrane Database Syst Rev. 2019;8(8):CD010233. |
| 27. | * Narratives Review Interest in allopurinol re-emerged in 1965 when it was recognised that it could be used to reduce uric acid levels in patients with gout and tumour related hyperuricemia [20, 26]. DOI: 10.1007/s10620-022-07719-x Study: weak evidence | Turbayne AK, Sparrow MP. Low-Dose Azathioprine in Combination with Allopurinol: The Past, Present and Future of This Useful Duo. Dig Dis Sci. 2022;67(12):5382-5391. |
| 28. | * Tierstudie / Experimentelle Arbeit mit Mäusen Purine nucleotides are vital for RNA and DNA synthesis, signaling, metabolism, and energy homeostasis. To synthesize purines, cells use two principal routes: the de novo and salvage pathways. Traditionally, it is believed that proliferating cells predominantly rely on de novo synthesis, whereas differentiated tissues favor the salvage pathway. Unexpectedly, we find that adenine and inosine are the most effective circulating precursors for supplying purine nucleotides to tissues and tumors, while hypoxanthine is rapidly catabolized and poorly salvaged in vivo. Notably, feeding mice nucleotides accelerates tumor growth, while inhibiting purine salvage slows down tumor progression, revealing a crucial role of the salvage pathway in tumor metabolism. These findings provide fundamental insights into how normal tissues and tumors maintain purine nucleotides and highlight the significance of purine salvage in cancer. DOI: 10.1016/j.cell.2024.05.011 Study: weak evidence | Tran DH, Kim D et al. De novo and salvage purine synthesis pathways across tissues and tumors. Cell. 2024;187(14):3602-3618.e20 |
| 29. | * narratives Review Self-renewal and differentiation are two characteristics of hematopoietic stem cells (HSCs). Under steady physiological conditions, most primitive HSCs remain quiescent in the bone marrow (BM). They respond to different stimuli to refresh the blood system. The transition from quiescence to activation is accompanied by major changes in metabolism, a fundamental cellular process in living organisms that produces or consumes energy. Byproducts from the cellular metabolism can also damage DNA. To counteract such insults, mammalian cells have evolved a complex and efficient DNA damage repair (DDR) system to eliminate various DNA lesions and guard genomic stability. In response to stress, HSCs mobilize out of the niche, entering the cell cycle for division [3]. The transition from quiescence to activation is accompanied by major metabolic and mitochondrial changes that are important for balanced decisions between self-renewal and differentiation to generate enough hematopoietic stem progenitor cells (HSPCs) while preventing HSC exhaustion. Fundamental cellular processes involved in metabolism can also damage DNA through increasing reactive oxygen species (ROSs) or generating toxic byproducts. It has emerged that cellular metabolic regulation not only generates DNA damage but also impacts DNA repair. Cellular metabolism is intimately linked to the maintenance of genomic integrity, with metabolic cues influencing DDR pathways and vice versa [5]. In general, the DNA damage in HSCs is endogenous, majorly induced by reactive oxygen species, aldehydes, and replication stress. Our recent study reveals that FA HSCs exhibit a heightened dependence on OXPHOS and undergo a rapid switch from glycolysis to OXPHOS under oxidative stress to cope with oxidative DNA damage. Mechanistically, the tumor suppressor p53 functions as the key master regulator mediating this transition. p53 regulates energy metabolism at the glycolytic and OXPHOS steps via the transcriptional regulation of its downstream genes, such as the synthesis of SCO2, a member of the COX-2 assembly involved in the electron-transport chain.
DOI: 10.3390/cells13090733 Study: weak evidence | Xu J, Fei P et al. Crosstalk between DNA Damage Repair and Metabolic Regulation in Hematopoietic Stem Cells. Cells. 2024;13(9):733. |
| 30. | ● Website | USDA and ODS-NIH Database: Purine Content of Foods Release 2.0 (2025). |
| 31. | ● Book: moderate evidence | Der Kleine Souci/Fachmann/Kraut, Lebensmitteltabelle für die Praxis. 6. Auflage. Stuttgart: Wissenschaftliche Verlagsgesellschaft; 2023. |
| 32. | * Originalarbeit (Primärstudie, experimentell-analytisch) 4. IN NUTRITIONAL THERAPY FOR GOUT AND HYPERURICEMIA In Japan, lifestyle interventions for patients with hyperuricemia/gout are shown in the Guidelines for the Management of Hyperuricemia and Gout.13) The major points of lifestyle interventions are nutritional therapy, restriction of alcohol consumption and recommendations for physical training. Nutritional therapy suggests an appropriate intake of energy and water and a reduced intake of dietary purines (less than 400 mg/d) and fructose. Education and proper guidance are also thought to play a crucial role in improving the clinical course of the disease with or without drug therapy in the United States.4,5,36,37) In the Japanese guidelines, food is categorized according to the purine amount (very large, large, small, very small). Patient with gout or hyperuricemia are advised to avoid overeating the foods that contain a very large or large amount of purine (Table 9). These foods contain >200 mg/100 g of purines. In several epidemiological studies, the consumption of meat and fish was strongly correlated with elevated uric acid serum concentrations and the risk of gout.6,8–12) In the present review, many meats and fishes contained >100 mg/100 g of purine.
Thus, foods containing small amounts of total purine and those containing mainly adenine and guanine are considered to be beneficial for hyperuricemic patients and those with gout.
5. CONCLUSION Two important points should be taken into consideration regarding nutritional therapy for gout or hyperuricemia with particular respect for the restriction of dietary purines. The first is the amount of total purines in the food and the second is what types of purine bases are included. The consumption of foods that contain >200 mg/100 g of purines, especially with a high ratio of hypoxanthine, is considered to be a high risk for hyperuricemia. This group includes animal meats, fish meats, and some shrimps. Epidemiological studies have shown that a high intake of such foods is related to the elevation of serum uric acid concentrations and thus is correlated with the risk of gout. Because liver and milt also contains a high amount of purines, heavy consumption of these foods is not recommended even if the purines are mainly adenine and guanine. However, moderate consumption of metallic fish that contain a considerable amount of guanine is recommended not only for the management of gout and hyperuricemia but also for decreasing cardiovascular disease risk. High intake of low-purine foodstuffs, such as dairy products, cereals, beans, vegetables, mushrooms, and soybean products, is strongly recommended. DOI: 10.1248/bpb.b13-00967 Study: weak evidence | Kaneko K, Aoyagi Y et al. Total purine and purine base content of common foodstuffs for facilitating nutritional therapy for gout and hyperuricemia. Biol Pharm Bull. 2014;37(5):709-721. |
| 33. | * Originalarbeit (Primärstudie, experimentell-analytisch) DOI: 10.1007/BF02023808 Study: weak evidence | Wolfram G, Colling M. Gesamtpuringehalt in ausgewählten Lebensmitteln. Zeitschrift für Ernährungswissenschaft. 1987;26:205-213. |
| 34. | ● Website | USDA United States Department of Agriculture. FoodData Central. |
| 35. | ● Website | Food Database (fddb info): Kalorientabelle und Ernährungstagebuch. |
| 36. | * Systematisches Review und Meta-Analyse Our systematic review and meta-analysis of prospective cohort studies supports the association between fructose intake and increased risk of developing gout. The strength of evidence for the association between fructose consumption and risk of gout was low, as assessed by GRADE. It means that further research is likely to have a significant impact on our confidence in the effect estimate and is likely to change the estimate. DOI: 10.1136/bmjopen-2016-013191 Study: strong evidence | Jamnik J, Rehman S et al. Fructose intake and risk of gout and hyperuricemia: a systematic review and meta-analysis of prospective cohort studies. BMJ Open. 2016;6(10):e013191. |
| 37. | * Prospektive Kohortenstudie In this cohort study of 122 679 US men and women, adherence to an overall plant-based dietary pattern that includes both healthy and unhealthy plant foods was not associated with gout. However, higher intake of a healthy plant-based diet that specifically emphasizes healthier plant-based foods was associated with lower gout risk, while an unhealthy plant-based diet was associated with higher gout risk, particularly in women. An overall plant-based diet index (PDI), as well as healthy (hPDI) and unhealthy (uPDI) versions of this index that emphasize healthy and less healthy plant-based foods, respectively. Supplement 1 -> foods in diets DOI: 10.1001/jamanetworkopen.2024.11707 Study: moderate evidence | Rai SK, Wang S et al. Adherence to Healthy and Unhealthy Plant-Based Diets and the Risk of Gout. JAMA Netw Open. 2024;7(5):e2411707. |
| 38. | * Prospektive Kohortenstudie Higher levels of meat and seafood consumption are associated with an increased risk of gout, whereas a higher level of consumption of dairy products is associated with a decreased risk. Moderate intake of purine-rich vegetables or protein is not associated with an increased risk of gout. DOI: 10.1056/NEJMoa035700 Study: moderate evidence | Choi HK, Atkinson K et al. Purine-rich foods, dairy and protein intake, and the risk of gout in men. N Engl J Med. 2004;350(11):1093-1103. |
| 39. | * Case-Crossover-Studie The study findings suggest that acute purine intake increases the risk of recurrent gout attacks by almost fivefold among gout patients. Avoiding or reducing amount of purine-rich foods intake, especially of animal origin, may help reduce the risk of gout attacks. We found that the short-term impact of purine from plant sources on the risk of gout attacks was substantially smaller than that from animal purine sources. Also, in a large prospective study of incident gout, the long-term, habitual consumption of purine-rich vegetables was not associated with the risk of incident gout. Interestingly, in that study, the highest quintile of vegetable protein consumption was actually associated with a 27% lower risk of gout compared with the lowest quintile. Our analysis of purine quantities suggests that these findings of small or null effects of purine intake from plant sources can be explained by the substantially lower amounts of purine content in those food items. Other healthy nutrients of vegetable items (eg, fibre or healthy fat) could contribute to reducing long-term weight gain and lowering insulin resistance. DOI: 10.1136/annrheumdis-2011-201215 Study: moderate evidence | Zhang Y, Chen C et al. Purine-rich foods intake and recurrent gout attacks. Ann Rheum Dis. 2012;71(9):1448-1453. |
| 40. | * Narratives Review Uric acid (UA) is produced in the liver and excreted through the kidneys and intestines. If UA is overproduced or its excretion reduces, the concentration of UA increases, leading to hyperuricemia and gout. The high concentration of UA is also related to cardiovascular disease, hypertension, obesity, and other diseases. Fruits are healthy foods. However, fruits contain fructose and small amounts of purine, and the product of their metabolism is UA. Therefore, theoretically, eating fruits will increase the concentration of serum UA. Fruit components are numerous, and their effects on serum UA are complex. According to the current research, fructose, purine, polyphenols, vitamin C, dietary fiber, and minerals present in fruits influence serum UA concentrations. . In most mammals, UA is oxidized to allantoin, which is easily soluble in water, under the action of enzyme UA oxidase. However, humans lack UA oxidase and hence cannot convert UA into allantoin (Wu et al., 2021). Therefore, excessive production or reduced excretion will increase the serum UA concentrations (Maiuolo et al., 2016). Hyperuricemia occurs when the serum UA exceeds its normal concentration. Persistent hyperuricemia can cause the deposition of UA crystals in joints and other places, leading to gout (Li et al., 2019). However, polyphenols can reduce the production of UA by inhibiting the activity of XO. Quercetin can bind to the active center of XO and prevent xanthine from entering the active center of XO; therefore, quercetin can inhibit the activity of XO, and reduce the generation of UA (Ahn et al., 2020; Mehmood et al., 2019). Additionally, gallic acid, epicatechin, catechin, hesperidin, naringenin, genistein, and other polyphenols are also effective XO inhibitors (Mehmood et al., 2019; Mohos et al., 2020). Figure 4 shows the mechanism of polyphenols inhibiting XO. Polyphenols can reduce UA levels by inhibiting XO, reducing the reabsorption of UA, and improving the excretion of UA. Vitamin C can not only reduce the serum UA levels, but also increase the antioxidant capacity of humans. Dietary fiber can slow down the reabsorption rate of UA and promote the excretion of UA. Minerals in fruits also reduce serum UA concentrations. DOI: 10.1111/jfbc.13911 Study: weak evidence | Zou F, Zhao X, Wang F. A review on the fruit components affecting uric acid level and their underlying mechanisms. J Food Biochem. 2021;45(10):e13911. |
| 41. | * Primärstudie, in vitro-Experimente / Laborstudie Various dietary flavonoids were evaluated in vitro for their inhibitory effect on xanthine oxidase, which has been implicated in oxidative injury to tissue by ischemia-reperfusion. Xanthine oxidase activity was determined by directly measuring uric acid formation by HPLC. The structure-activity relationship revealed that the planar flavones and flavonols with a 7-hydroxyl group such as chrysin, luteolin, kaempferol, quercetin, myricetin, and isorhamnetin inhibited xanthine oxidase activity at low concentrations (IC50 values from 0.40 to 5.02 μM) in a mixed-type mode, while the nonplanar flavonoids, isoflavones and anthocyanidins were less inhibitory. These results suggest that certain flavonoids might suppress in vivo the formation of active oxygen species and urate by xanthine oxidase. DOI: 10.1271/bbb.63.1787 Study: weak evidence | Nagao A, Seki M, Kobayashi H. Inhibition of xanthine oxidase by flavonoids. Biosci Biotechnol Biochem. 1999;63(10):1787-1790. |
| 42. | * Narratives Review The SLC28 family consists of three subtypes of sodium-dependent, concentrative nucleoside transporters, CNT1, CNT2, and CNT3 (SLC28A1, SLC28A2, and SLC28A3, respectively), that transport both naturally occurring nucleosides and synthetic nucleoside analogs used in the treatment of various diseases. These subtypes differ in their substrate specificities: CNT1 is pyrimidine-nucleoside preferring, CNT2 is purine-nucleoside preferring, and CNT3 transports both pyrimidine and purine nucleosides. Early studies in isolated mammalian tissues and cell lines demonstrated that nucleoside uptake is characterized by low- and high-affinity systems and that the high-affinity system(s) is active, concentrative, and Na+-dependent. The low-affinity system is now recognized as the equilibrative nucleoside transporter (ENT) family, SLC29, whereas SLC28 is responsible for high-affinity transport. DOI: 10.1007/s00424-003-1107-y Study: weak evidence | Gray JH, Owen RP, Giacomini KM. The concentrative nucleoside transporter family, SLC28. Pflugers Arch. 2004;447(5):728-734. |
| 43. | ● Most patients with nephrolithiasis (75%-85%) form calcium stones, most composed primarily of calcium oxalate (monohydrate or dihydrate) or calcium phosphate. The other main types include uric acid (8%-10%), struvite (calcium magnesium ammonium phosphate, 7%-8%), and cystine stones (1%-2%). Calcium oxalate stones are the most common type of renal calculi, comprising 70% to 75% of all urinary stones. While chemically identical, they may present as 2 different crystalline forms: calcium oxalate monohydrate (whewellite, very hard) or a dihydrate (weddelite, brittle). These stones typically form in acidic urine but may be found with calcium phosphate, forming the central nidus.
Calcium phosphate calculi may be seen as the less soluble carbonate apatite (hydroxyapatite, apatite) and brushite (calcium hydrogen phosphate). They account for about 10% of all renal calculi. Hydroxyapatite is more commonly found than brushite and is the calcium salt that forms bone. In general, calcium phosphate stones tend to grow faster and larger than calcium oxalate calculi. These stones are off-white, grayish-white, or yellowish in color. Calcium phosphate stones form in alkaline urine and are typically associated with abnormal metabolic factors, such as hyperparathyroidism and renal tubular acidosis. Uric acid calculi only form in acidic urine, usually with a pH less than 5.5. This acid is the most common composition of bladder stones and is typically radiolucent. Uric acid accounts for 8% to 10% of urinary calculi, and the incidence is increasing worldwide. This condition is most closely associated with diabetes, morbid obesity, metabolic syndrome, and older age at presentation. This is the only kidney stone that can be reasonably expected to dissolve if the urinary pH is sufficiently elevated and maintained. This type of stone is also more likely to form from excessive urinary acidity rather than hyperuricosuria. Uric acid stones may be yellow, orange, reddish, or brown, depending on the amount of blood-derived pigment they may have accumulated. Preventive treatment involves urinary alkalinization and possibly allopurinol if there is hyperuricosuria. Struvite or triple phosphate (calcium, ammonium, magnesium phosphate) stones are always associated with infection and increased pH levels. They frequently form staghorn stones and comprise 7% to 8% of all urinary calculi worldwide. Struvite stones are caused by the action of urease from bacteria, which increase the urinary pH and generate ammonia, leading to triple phosphate precipitation and stone formation. To treat the infection adequately, complete elimination of all stone material is necessary. Struvite stones appear chalky, white, or grayish. Their surface is usually smooth and relatively brittle, as they can be broken relatively easily. Cystine stones are caused by an uncommon familial genetic defect and account for only 1% to 2% of all urinary stones. They tend to be amber, tan, or yellowish in color with a waxy appearance. Cystine stones may turn somewhat greenish after exposure to air. The stones are not calcified but resistant to shockwave therapy; therefore, laser lithotripsy is usually the preferred treatment. Preventive treatment includes very high levels of hydration (>3 liters of urine/day), urinary alkalinization to a pH of 7.5 or more, and tiopronin, a reducing compound, if necessary. DOI: - Website | Leslie SW, Sajjad H, Murphy PB. Renal Calculi, Nephrolithiasis. Treasure Island: StatPearls Publishing; 2025. |
| 44. | * Primärstudie: Laborstudien The total purine contents of 100 mL of plain soymilk, 100 mL of adjusted soymilk, 100 mL of low-fat milk, and 100 mL of normal milk were 19.34 ± 0.43, 3.47 ± 0.06, 0.15 ± 0.03, and 0.14 ± 0.01 mg, respectively. DOI: 10.1080/15257770.2022.2093362 Study: weak evidence | Fukuuchi T, Itahashi I, et al. Determination of total purine and free purine content in milk, soymilk, and enteral nutritional supplements to assist nutritional therapy for hyperuricemia and gout. Nucleosides Nucleotides Nucleic Acids. 2022;41(12):1287-1295. |
| 45. | * Primärstudie, experimentell: Tiermodell und In-Vitro-Experimente Fructose consumption is a potential risk factor for hyperuricemia because uric acid (UA) is a byproduct of fructose metabolism caused by the rapid consumption of adenosine triphosphate and accumulation of adenosine monophosphate (AMP) and other purine nucleotides. Additionally, a clinical experiment with four gout patients demonstrated that intravenous infusion of fructose increased the purine de novo synthesis rate, which implied fructose-induced hyperuricemia might be related to purine nucleotide synthesis.
In liver, fructose can be metabolized more readily than glucose because of a specific enzyme (fructokinase), which catalyzes the conversion of fructose to fructose-1-phosphate using adenosine triphosphate (ATP) as a phosphate donor (8). Fructokinase is not regulated and phosphorylates fructose as rapidly as it can, leading to depletion of intracellular ATP to generate adenosine monophosphate (AMP). AMP accumulation stimulates AMP deaminase, which results in degradation of purine nucleotide (PNs) to UA, and increases the serum UA level (9, 10): this is a well-known mechanism of fructose-induced hyperuricemia. DOI: 10.3389/fnut.2022.1045805 Study: weak evidence | Zhang P, Sun H, et al. Dietary intake of fructose increases purine de novo synthesis: A crucial mechanism for hyperuricemia. Front Nutr. 2022;9:1045805. |
| 46. | ● Normbereich: 1.003-1.030 g/mL (eventuell höher). Werte der Dichte:
Website | Universität Bern: Dichte (spezifisches Gewicht) und Osmolalität. UroSurf. 2017. |
| 47. | * Primärstudie: Kombination aus Tierexperimenten (gnotobiotische Mäuse) und mikrobiologischen Kulturen. Bezieht zusätzlich eine Human-Kohorte mit Serum-Harnsäure und Koronarverkalkung ein. We asked to what extent UA could serve as a source of carbon and energy for gut bacteria, and to what extent the gut microbiota composition might affect host systemic purine concentrations.
These results again suggested that the gut microbiome modulates abundance of purines both in the gut and systemically and was the impetus for attempts to isolate anaerobic purine-degrading bacteria (PDB).
Our results showing that PDB lower the abundance of some purines in the intestine (Fig. 5) suggest that these organisms may lower circulating UA levels by decreasing the burden of purines bioavailable to the host.
In summary, the work presented here shows that anaerobic purine utilization is widespread among gut-dwelling bacteria and suggests that microbial purine degraders are important modulators of host purine homeostasis in the gut and of UA levels in circulation.
Thus, gut microbes are important drivers of host global purine homeostasis and serum uric acid levels, and gut bacterial catabolism of purines may represent a mechanism by which gut bacteria influence health.
Altogether, these results (i) suggest that phylogeny is a poor predictor of microbial purine utilization; (ii) indicate that the presence of the identified genes does not correlate with the breadth of purines utilized by an organism; (iii) demonstrate effects on purine metabolism of two nutritional parameters–i.e., carbon source and metals availability; and (iv) underscore the need for assessments beyond genomics when making predictions about purine metabolism by the gut microbiota. DOI: 10.1016/j.chom.2023.05.011 Study: weak evidence | Kasahara K, Kerby RL, et al. Gut bacterial metabolism contributes to host global purine homeostasis. Cell Host Microbe. 2023;31(6):1038-1053.e10. |
| 48. | * Primärstudie: Kombination aus mikrobiologischen Kulturen, Genom- und Transkriptomanalysen, Tiermodellen (uricase-defiziente Mäuse) sowie Human-Daten. Uric acid is an intermediate in purine degradation in mammals. In most mammals, uric acid is converted to freely soluble allantoin via urate oxidase (uricase), which is then excreted via the kidney.
Here, we find that anaerobic uric acid metabolism is widespread among members of the human gut microbiome, occurring in ∼1/5 of bacteria from 4 of 6 major phyla.
In contrast to aerobic pathways that rely on oxygen-dependent uricase to initiate uric acid metabolism, we find that anaerobic pathways break down uric acid through action of uncharacterized ammonia lyase, peptidase, carbamoyl transferase, and oxidoreductase enzymes. The genes encoding these enzymatic functions map to a conserved gene cluster that is broadly distributed across distantly related bacterial taxa and are required for anaerobic uric acid metabolism to lactate and SCFAs.
However, the uric acid genes identified in our study are highly predictive of uric acid metabolism activity in gut bacteria, indicating that this gene cluster encodes a predominant pathway for anaerobic uric acid metabolism in the gut. A recent study also identified uric-acid-degrading gut bacteria, the same set of genes, and demonstrated that gut bacteria influence uric acid levels in the host, thus reinforcing our conclusions.
To address whether microbiota depletion influences fecal uric acid levels, we re-analyzed metabolomics data from the Food and Resulting Microbial Metabolites (FARMM) study exploring the role of diet in microbiome metabolite recovery after disruption with antibiotics and polyethylene glycol. We found that microbiota depletion resulted in dramatically elevated fecal levels of uric acid (Figure S7A). Fecal uric acid levels rapidly returned to baseline in subjects fed a vegan or omnivore diet, but those fed a fiber-free synthetic diet (exclusive enteral nutrition; EEN) showed a protracted recovery, with persistent elevations of fecal uric acid throughout the recovery phase (Figure S7A).
These results suggest that a lack of dietary fiber following microbiome perturbation imparts a sustained dysregulation of uric acid metabolism in the gut. DOI: 10.1016/j.cell.2023.06.010 Study: weak evidence | Liu Y, Jarman JB, et al. A widely distributed gene cluster compensates for uricase loss in hominids. Cell. 2023;186:3400–3413.e20. |
| 49. | * Narratives Review Gout, a prevalent and painful metabolic disease often associated with obesity and aging, is caused by the deposition of urate crystals in joints, bones, or soft tissues1. Urate is an intermediate metabolite within the purine degradation pathway, predominantly derived from uric acid under physiological pH levels.
Hyperuricemia occurs due to excessive uric acid production or insufficient excretion, which is associated with various chronic diseases, including type 2 diabetes, chronic kidney disease, cardiovascular disorders, and metabolic syndrome. DOI: 10.1016/j.apsb.2023.11.013 Study: weak evidence | Wang L, Ye J. Commentary: Gut microbiota reduce the risk of hyperuricemia and gout in the human body. Acta Pharm Sin B. 2024;14(1):433-435. |
| 50. | * Primärstudie: Tierstudie Probiotics, especially the LAB, have been widely used in the manufacture of dairy products such as yogurt, cheese, and pickled vegetables. Increasing evidence underscores the beneficial effects of the lactic acid bacteria on human physiology and pathology. Among the most distinctive benefits of Lactobacillus is protection against chronic disease hyperuricemia.
Collectively, our work provides substantial evidence identifying the specific role of L. plantarum in improvement of urate circulation. We highlight the importance of the enzymes RihA–C existing in L. plantarum for the urate metabolism in hyperuricemia mice induced by a high-nucleoside diet. Although the direct connection between nucleobase transport and host urate levels has not been identified, the lack of nucleobase transporter in intestinal epithelial cells might be important to decrease its absorption and metabolization for urate production, leading to the decrease of serum urate in host. These findings provide important insights into urate metabolism regulation. DOI: 10.1186/s40168-023-01605-y Study: weak evidence | Li M, Wu X, et al. Lactiplantibacillus plantarum enables blood urate control in mice through degradation of nucleosides in gastrointestinal tract. Microbiome. 2023;11(1):153. |
| 51. | * Beobachtungsstudie mit kontrollierter Intervention (kontrollierte Fütterungsstudie, experimentell-analytisch) Thirty-one healthy volunteers between the ages of 18 and 60 were included in the study, however one withdrew before completing the protocol. As a result, 30 are included in the analysis, 10 in each group. Since gut microbiota metabolites are influenced by diet, we performed a longitudinal analysis of the impact of three divergent diets, vegan, omnivore, and a synthetic enteral nutrition (EEN) diet lacking fiber, on the human gut microbiome and its metabolome, including after a microbiota depletion intervention. Omnivore and vegan, but not EEN, diets altered fecal amino acid levels by supporting the growth of Firmicutes capable of amino acid metabolism. This correlated with relative abundance of a sizable number of fecal amino acid metabolites, some not previously associated with the gut microbiota. The effect on the plasma metabolome, in contrast, were modest. The impact of diet, particularly fiber, on the human microbiome influences broad classes of metabolites that may modify health.
DOI: 10.1016/j.chom.2020.12.012 Study: moderate evidence | Tanes C, Bittiner K, et al. Role of dietary fiber in the recovery of the human gut microbiome and its metabolome. Cell Host & Microbe. 2021;29(3):P394-P407.E5. |
| 52. | * Narratives Review In addition to its role as a byproduct of purine metabolism, uric acid is recognized for its multifaceted effects, which include antioxidant, pro-oxidant, pro-inflammatory, nitric oxide regulation, immune system interactions, and anti-aging properties.
In recent years, studies have revealed that UA activates the TLR4-NLRP3 inflammatory complex, which is a multi-protein complex that plays a pivotal role in initiating the innate immune response to various danger signals, including MSU crystals. Upon recognition of MSU crystals, the NLRP3 inflammasome is activated, leading to the cleavage of pro-inflammatory cytokines, specifically interleukin-1β (IL-1β) and interleukin-18 (IL-18).
Hyperuricemia, by inducing oxidative stress and inflammation, diminishes the expression of eNOS and the synthesis of NO, while elevating levels of inflammatory cytokines such as IL-6 and TNF-α, ultimately impairing endothelial function. In addition, NO is involved in inhibiting platelet aggregation, leukocyte adhesion, and inflammation. It also contributes to various signaling pathways that affect cardiac function, nerve conduction, and the immune response. The interaction between uric acid and NO is bidirectional. When concentrations are low, uric acid acts as a natural antioxidant that scavenges free radicals and prevents oxidative damage.
Dietary selections abundant in purine, particularly nucleic acids, notably contribute to the production of uric acid.
Consumption of purine-rich meats such as beef, pork, lamb, and seafood like oysters, shrimp, and tuna, as well as dietary fructose, are known to elevate uric acid (UA) production. Additionally, alcohol metabolism from beer and distilled spirits, along with certain medical conditions such as tumor lysis syndrome and obesity, pose increased risks for hyperuricemia.
Conversely, protein intake from either animal or plant sources demonstrated a contrasting impact on the prevalence of hyperuricemia.
Increased levels of uric acid result in inflammation and oxidative stress, which serve as potential risk factors for cellular senescence, apoptosis, and disruptions in the cell cycle. Conversely, physiological concentrations of uric acid (5 mg/dl) exhibit anti-aging effects by enhancing growth factor activity in aging cells. However, at higher concentrations (10 mg/dl), uric acid promotes cellular senescence and downregulates EGF/EGFR signaling. DOI: 10.1038/s41392-024-01916-y Study: weak evidence | Du L, Zong Y, et al. Hyperuricemia and its related diseases: mechanisms and advances in therapy. Signal Transduct Target Ther. 2024;9(1):212. |
| 53. | * Primärstudie, experimentell: Laborstudie Fadenwurm (Tiermodell) The process of aging has fascinated humankind for thousands of years. Aging has been defined as a synchronous global decline in physiological and psychological function, accompanied by many diseases, including type 2 diabetes, cancer and hypertension. One of the main mechanisms underlying aging and age-associated disease is a chronic elevation of reactive oxygen species (ROS). Reactive oxygen species (ROS) are generated as a byproduct of normal metabolism and are thought to be produced mainly in mitochondria. ROS have been increasingly recognized as a pivotal mediator of several oxidative stress responses, and an imbalance between ROS production and elimination has been considered a risk factor for aging and a number of age-related diseases. In this work, we investigated the impact of uric acid as an antioxidant on the health span and life span of nematode C. elegans. Our results from this study indicated that uric acid significantly extended the life span, delayed age-related physiological functions, and enhanced oxidative stress resistance in C. elegans by activating the stress-related transcription factors DAF-16/FOXO and SKN-1/NRF2 and by regulating the insulin/IGF-1 signaling (IIS) and reproductive signaling pathways. These results reveal that purine metabolic intermediates play an important role in the regulation of aging and that endogenous purine metabolites may be developed into potential strategies for the prevention and treatment of aging and age-related diseases. These studies of uric acid suggest that, due to the antioxidative activity of uric acid, higher concentrations of uric acid are generally beneficial compared with lower concentrations, but higher levels that result in crystal formation are detrimental. Therefore, in future research, our goal is to further clarify the molecular mechanism of uric acid regulation of life span and to determine the appropriate concentration that is beneficial to the health of the body. In addition, we find that mitochondrial function plays an important role in uric acid-mediated life span extension. DOI: 10.18632/aging.102781 Study: weak evidence | Wan QL, Fu X, et al. Uric acid induces stress resistance and extends the life span through activating the stress response factor DAF-16/FOXO and SKN-1/NRF2. Aging (Albany NY). 2020;12(3):2840-2856. |
| 54. | * Narratives Review Purines are essential organic compounds widely present in biological organisms in various forms, including free purines, nucleosides, and nucleotides. They enter the human body mainly through dietary intake, with foods classified into high, moderately high, moderately low, and low-purine categories based on purine content. While purines play vital physiological roles in genetic information storage, energy transfer, and signal transduction, excessive accumulation of uric acid (UA), the final metabolite of purine degradation, can lead to health issues such as gout and kidney stones. Thus, managing dietary purine intake is critical for preventing related diseases.
In recent years, with shifting dietary patterns and lifestyle changes, health issues related to high-purine diets have become increasingly prominent, emerging as a critical public health concern worldwide.
The purine content in foods varies significantly—organ meats, seafood, and meat are particularly rich in purines, whereas vegetables, fruits, and dairy products contain relatively lower levels.
To effectively mitigate the health risks associated with high-purine diets, regulating dietary purine intake has become a key preventive strategy. Studies have shown that adopting a well-balanced diet, reducing the consumption of purine-rich foods, and implementing lifestyle modifications—such as increasing water intake, maintaining a healthy weight, and limiting alcohol consumption—can significantly lower UA levels and reduce the risk of gout and other purine-related diseases. DOI: 10.1016/j.tifs.2025.105191 Study: weak evidence | Song Y, Li Q, et al. Dietary purines and health: Metabolism, impact, and regulation. Trends in Food Science & Technology. 2025;163:105191. |
| 55. | * Primärstudie mit Daten von zwei Kohortenstudien Serum uric acid (SUA), a byproduct of purine metabolism, exerts both antioxidant and pro-inflammatory effects, making its role in aging and chronic diseases a subject of ongoing debate. Despite this, the mechanisms by which SUA influences the aging process remain poorly understood. Serum uric acid (SUA), the end product of purine metabolism, has emerged as a particularly contentious factor in aging research. On one hand, SUA functions as an evolutionarily conserved antioxidant capable of scavenging reactive oxygen species (ROS) (6). On the other, elevated SUA levels can activate the NLRP3 inflammasome (7), impair endothelial function, and are linked to hypertension (8), chronic kidney disease (CKD) (9), and cardiovascular events. This biological paradox has been reflected in epidemiologic studies, many of which describe a U-shaped association between SUA levels and mortality risk (10, 11). Nevertheless, the mechanisms driving this nonlinear relationship remain unclear. Additionally, prior research has predominantly focused on single aging biomarkers or ethnically homogeneous populations, limiting both mechanistic insight and generalizability. To address these gaps, we conducted a comparative analysis leveraging data from two nationally representative cohorts: the National Health and Nutrition Examination Survey (NHANES, 1999–2010) in the United States and the China Health and Retirement Longitudinal Study (CHARLS, 2011–2015). We applied three complementary biological aging measures—Klemera–Doubal Method Biological Age (KDM-BA), Phenotypic Age (PhenoAge), and Allostatic Load (AL)—to evaluate the associations between SUA, biological aging, and mortality outcomes. Based on data from the NHANES and CHARLS cohorts, we found that elevated SUA levels were significantly associated with accelerated biological aging in both populations. In the NHANES cohort, higher SUA levels were also linked to an increased risk of all-cause and premature mortality, with a U-shaped nonlinear relationship. However, this association was not observed in the CHARLS cohort, suggesting potential population-specific differences. These findings underscore the role of SUA as a potential contributor to aging and mortality risk, highlighting the need for further research to clarify the causal relationship and evaluate the long-term benefits and risks of uric acid-lowering strategies. Elevated SUA is associated with accelerated biological aging in both U.S. and Chinese populations, but its link to mortality was evident only in the NHANES cohort. These findings highlight SUA as a potential aging marker and call for further population-specific investigation.
DOI: 10.3389/fnut.2025.1569798 Study: moderate evidence | Zhao C, Zhao L, et al. The impact of serum uric acid on biological aging and mortality risk: insights from the NHANES and CHARLS cohorts. Front Nutr. 2025;12:1569798. |
| 56. | * Narratives Review As one of the four major macromolecules (percentage weight in mammalian cell: DNA, ∼7 pg, 0.3%; RNA, ∼20 pg, 1%; protein, ∼500 pg, 20%; and polysaccharide, ∼2 μg, 78.7%. DOI: 10.1016/j.gpb.2014.04.002 Study: weak evidence | Wu J, Xiao J, et al. Ribogenomics: the science and knowledge of RNA. Genomics Proteomics Bioinformatics. 2014;12(2):57-63. |
| 57. | * Pathogenese der Gicht und diätetische Einflüsse Hauptverantwortlich für die Gicht ist die akkumulierte Harnsäuremenge im Körper. Diese ergibt sich aus dem Gleichgewicht von diätetischer Zufuhr, körpereigener Synthese und renaler Ausscheidung. Eine Hyperurikämie resultiert aus einer Überproduktion von Harnsäure, einer verminderten renalen Ausscheidung der Harnsäure oder häufig aus einer Kombination von beidem (12, 13). Früher wurde angenommen, dass man hauptsächlich den exogenen Harnsäureanteil diätetisch beeinflussen kann. Inzwischen weiss man aber, dass sich diätetisch mehr erreichen lässt durch Beeinflussung der endogenen Harnsäuresynthese und der renalen Harnsäureausscheidung (14). Diätetische Massnahmen bei Gicht haben drei Ziele 1. Zur Schubprophylaxe sollen diäte tische Auslöser vermieden werden (Abbildung 3). Dazu gehören üppige Fleischmahl - zeiten, Innereien, Meeresfrüchte, Alkohol und fruktosehaltige Nahrung/Getränke. Diese erhöhen akut den Serumharnsäurespiegel, was Arthritisschübe provoziert (15, 16). Zudem können zur Schubprophylaxe entzündungshemmende Nahrungsbestand teile eingenommen werden wie beispielsweise Milchextrakte oder Kirschen (17, 18). 2. Die Diät soll längerfristig die Harnsäurekonzentration im Blut beziehungsweise die Harnsäuremenge im Körper reduzieren, was das Risiko für Gichtmanifesta - tionen senkt. Gicht: Die traditionelle purinarme Diät ist out Die Gichtdiät darf sich nicht darauf beschränken, nur die Serumharnsäure und die Schubhäufigkeit zu senken, sondern sie muss auch das häufig assoziierte metabolische Syndrom und das erhöhte kardiovaskuläre Risiko angehen. Die traditionelle purinarme Diät kann nicht mehr empfohlen werden. Insbesondere ist eine proteinreiche pflanzliche Kost trotz ihres hohen Puringehalts sogar günstig. In erster Linie ist eine Normalisierung des Körpergewichts durch eine langsame Gewichtsreduktion mittels Kalorienrestriktion und vermehrter körperlicher Aktivität anzustreben. Die Zufuhr von Fleisch, Innereien und Meeresfrüchten sollte eingeschränkt werden. Bier ist strikt zu meiden, und auch auf Spirituosen sollte möglichst verzichtet werden. Regelmässiges Trinken von wenig Wein ist hingegen akzeptabel und hinsichtlich des kardiovaskulären Risikos günstig. Der Konsum von Milchprodukten reduziert das Gichtrisiko. Auch Kaffeekonsum und Vitamin-C-Supplemente wirken präventiv. Adrian Forster ADRIAN FORSTER1, ANDREAS KREBS2 1Klinik St. Katharinental, Diessenhofen 2Praxis für Rheumatologie, Kloten, und Rheumaklinik, Universitätsspital Zürich Andreas Krebs ENTZÜNDLICHE RHEUMATISCHE ERKRANKUNGEN UND ERNÄHRUNG 21 1/13 3. Die Gicht geht häufig mit Adipositas, arterieller Hypertonie, verminderter Glukosetoleranz und Hyperlipidämie als Komorbiditäten einher. Gichtpatienten haben ein stark erhöhtes kardiovaskuläres Risiko und dadurch eine erhöhte Sterblichkeit (19, 20). Die Diät soll deswegen diesen Komorbiditäten (metabolisches Syndrom) und dem kardiovaskulären Risiko entgegenwirken (21, 22). Gewisse diätetische Massnahmen zur Harnsäuresenkung (z.B. Verzicht auf Früchte und fetthaltige Fische) sollten Gichtpatienten nur in Ausnahmefällen empfohlen werden, weil sie hinsichtlich des kardiovaskulären Risikos ungünstig sind (1) Probleme der traditionellen purinarmen Diät Die herkömmliche Diätempfehlung bei Gicht war, auf Nahrungsmittel und Getränke zu verzichten, die Gichtattacken provozieren können, wie zum Beispiel üppige fleischhaltige Mahlzeiten mit viel Bier. Vor allem aber wurde eine konsequente purinarme Kost empfohlen. Diese hat sich jedoch als wenig wirksam erwiesen (nur selten Harnsäuresenkungen um über 60 µmol/l erzielbar) und wird ausserdem von den meisten Patienten nur schlecht akzeptiert (23). Zudem enthält eine purinarme Ernährung oft viele raffinierte Kohlenhydrate (einschliesslich Fruktose) und gesättigte Fette. Diese verstärken die Insulinresistenz und erhöhen das Risiko für ein metabolisches Syndrom und seine Komplikationen (23). Umgekehrt kann eine Diät, die eine Verminderung der Insulinresistenz anstrebt, nicht nur einem metabolischen Syndrom entgegenwirken und das kardiovaskuläre Risiko reduzieren, sondern auch den Serumharnsäurespiegel senken; ein Beispiel dafür ist die mediterrane Diät (24). Entsprechend gehören heute eine Gewichtsabnahme, ein vermehrter Konsum von Milchprodukten und eine eingeschränkte Fruktosezufuhr zu den wichtigsten Diätempfehlungen bei Gicht (1, 14, 21, 22). Die Annahme, dass eine proteinund purinreiche pflanzliche Ernährung ungünstig ist, hat sich als falsch erwiesen (2, 3). Die klassische purinarme Diät kann deswegen heute nicht mehr empfohlen werden (1). Langsame Gewichtsreduktion Bei Übergewicht und Adipositas ist eine langsame Gewichtsreduktion anzustreben, weil erhöhte Serumharnsäurespiegel und ein erhöhtes Gichtrisiko damit assoziiert sind (1, 4, 5, 21, 22). Für Gichtpatienten eignen sich Fastenkuren nicht, weil diese aufgrund der Ketoazidose Anfälle provozieren können. Selbstverständlich ist auch vermehrte körperliche Aktivität zur Gewichtsreduktion zu empfehlen (1). Narratives Review DOI: - Study: weak evidence | Forster A, Krebs A. Gicht – die traditionelle purinarme Diät ist out. Schweizerische Zeitschrift für Ernährungsmedizin. 2013;1:20-24. |
| 58. | * Erhöhte Harnsäurewerte im Blut spielen pathophysiologisch sowohl bei der Inzidenz als auch Rekurrenz von Gichtanfällen eine wichtige und kausale Rolle. Des Weiteren bestehen bei Patienten mit Gicht häufig Komorbidiäten wie z.B. Diabetes mellitus, arterielle Hypertonie oder koronare Herzkrankheit. Interessanterweise konnte gezeigt werden, dass unter einer harnsäuresenkenden Therapie mit Allopurinol eine akute Zufuhr von purinreichen Lebensmitteln einen unabhängigen Risikofaktor für einen Gichtanfall darstellt (10). Es sollte jedoch nicht unerwähnt bleiben, dass lediglich 25 Prozent der Personen mit einer Hyperurikämie bis 595 umol/l (=10 mg/dl) innerhalb von 10 Jahren einen Gichtanfall entwickeln (13). Schlussfolgerung
Narratives Review DOI: - Study: weak evidence | Mohebbi N. Einfluss von Ernährung auf die Harnsäure– Wahrheit oder Mythos in Fakten. Schweizer Zeitschrift für Ernährungsmedizin. 2020;2:14-17. |
| 59. | * The clinical symptoms of gout develop in several stages, including asymptomatic hyperuricemia, MSU crystal formation, intermittent gout and chronic gout. While current clinical principles based on medicinal management for gout have been well implemented, dietary modification and lifestyle changes have also been recommended for gout patients, since a suboptimal diet and obesity/diabetes-diseases of affluence contribute significantly to the risk of developing gout, increasing the burden of medical expenses. Purine source analyses show that nearly two-thirds of purines in the body are endogenous, and the remaining purines that enter the body via foods are known as exogenous purines. Thus, overindulgent intake of a high-purine diet, including seafoods and animal offal, can trigger the excessive accumulation of purine metabolites, giving rise to the excessive accumulation of uric acid in the body. In addition, some purine-free drinks can accelerate the promotion of purine degradation; for example, alcohol intake consumes large amounts of ATP to produce AMP in the liver, leading to the rapid occurrence of increased SUA levels. The consumption of yeast-rich foods, such as bread and yeast drinks, can lead to a high colonization of Saccharomyces cerevisiae in the gut, which can gradually elevate the secretion of uric acid in the host. The associations between diet-induced gut microbiota reconstruction and the progression of hyperuricemia/gout have been highlighted in recent research, as evidenced by the fact that long-term adherence to the typical Western diet caused an obvious reduction in the diversity of the gut microbiota, particularly those that degrade uric acid and produce metabolites known to benefit uric acid excretion. It has been shown that the beneficial dietary patterns against hyperuricemia usually contain a higher intake of vitamins, fiber, and unsaturated fatty acids and are often supplemented with appropriate amounts of minerals and high-quality protein, promoting a health state in which systemic metabolism is prone to disease improvement. It has been reported that overweight/obesity was connected with 60% of hyperuricemia cases in a clinical trial of 14,624 adults, possibly due to lipid metabolic disorder promoting purine metabolism by elevating XO activity. In addition, high dietary protein intake can also affect uric acid homeostasis, since protein digestion can generate several amino acids, such as glutamine, glycine and threonine, to induce purine synthesis, promoting the development of hyperuricemia. Narratives Review DOI: 10.3390/nu14173525 Study: weak evidence | Zhang Y, Chen S, et al. Gout and diet: a comprehensive review of mechanisms and management. Nutrients. 2022;14(17):3525. |
| 60. | * RCT DOI: 10.3389/fnut.2025.1634215 Study: strong evidence | Kahleova H, Maracine C, et al. Dietary acid load on the Mediterranean and a vegan diet: a secondary analysis of a randomized, cross-over trial. Front Nutr. 2025;12:1634215. |
| 61. | * Querschnittstudie DOI: 10.1053/j.jrn.2008.04.007 External Link Study: moderate evidence | Ausman LM, Oliver LM, et al. Estimated Net Acid Excretion Inversely Correlates With Urine pH in Vegans, Lacto-Ovo Vegetarians, and Omnivores. Journal of Renal Nutrition. 2008;18(5):456–465 |
| 62. | * Narratives Review DOI: 10.3390/nu11081736 Study: weak evidence | Jakše B, Jakše B, et al. Uric Acid and Plant-Based Nutrition. Nutrients. 2019;11(8):1736. |
| 63. | * Querschnittstudie DOI: 10.1093/jn/nxx003 Study: moderate evidence | Esche J, Krupp D, Mensink GBM, Remer T. Dietary Potential Renal Acid Load Is Positively Associated with Serum Uric Acid and Odds of Hyperuricemia in the German Adult Population. J Nutr. 2018;148(1):49-55. |
| 64. | Ferraro PM, Curhan GC. Serum Uric Acid and Risk of Kidney Stones. Am J Kidney Dis. 2017;70(2):158-159. | |
| 65. | * Kohortenstudie DOI: 10.1093/rheumatology/keaf091 Study: moderate evidence | Wang C, Guo K, et al. Impact of adding urine alkalization therapy to xanthine oxidase inhibitor in gout management: a prospective cohort study. Rheumatology (Oxford). 2025;64(6):3509-3517. |
| 66. | * Cross-Over-Studie DOI: 10.1186/1475-2891-11-39 Study: moderate evidence | Kanbara A, Miura Y, Hyogo H, Chayama K, Seyama I. Effect of urine pH changed by dietary intervention on uric acid clearance mechanism of pH-dependent excretion of urinary uric acid. Nutr J. 2012;11:39. |
| 67. | * Narratives Review DOI: 10.1007/s00424-024-02910-7 Study: weak evidence | Wieërs MLAJ, Beynon-Cobb B, et al. Dietary acid load in health and disease. Pflugers Arch. 2024;476(4):427-443. |
| 68. | * A higher level of PRAL was found to be associated with odds of hyperuricemia. There was a positive association between elevated NEAP and hyperuricemia. According to previously studies [21], PRAL and NEAP were calculated by the following formulas, respectively:
According to previous reports, high dietary acid load is characterized by both a high consumption of acid precursors and a low intake of base precursors [21]. Acid precursors are mainly from phosphorus and proteins, which have rich sulfur-containing amino acids, while base precursors are largely derived from potassium, magnesium, calcium etc. [23] The aforementioned reveals that high dietary acid load may attribute to a diet pattern with rich in animal-derived food but low in plant-derived food. Taking the diet pattern with high acid load in a long term might change the acid–base balance to acidosis [28]. Even though the potential mechanisms involved in dietary acid load and the risk of hyperuricemia is indeterminate, a previous study indicated that an alkaline urine pH would help excrete uric acid excretion, but not an acidic urine pH [29]. Other studies also exhibited a positive relationship between higher dietary acid load and risk of hyperuricemia. Dayeon et al. [29], found a 21% higher risk for hyperuricemia in middle-aged and older Korean adults with the highest quartile of PRAL, and a 17% higher risk in NEAP according to the Korean Genome and Epidemiology Study [29]. Another cross-sectional study conducted in German and with 6894 adults also identified a significant association between higher dietary acid load and risk of hyperuricemia [30, 31]. The German adults with lower PRAL were found to have a lower odd for hyperuricemia (OR: 0.60; 95% CI: 0.43, 0.83). Our study was in accord with these findings [30, 31]. Even though these studies were observational design, the effect of the higher dietary acid load on chronic diseases cannot be ignored. Fall-Kontroll-Studie DOI: 10.1186/s12902-022-01192-3 Study: moderate evidence | Shao SS, Lin CZ, et al. Higher dietary acid load is associated with hyperuricemia in Chinese adults: a case-control study. BMC Endocrine Disorders. 2022;22:286. |
| 69. | * This study showed a significant positive association between two indicators of dietary acid load (PRAL, and DAL) and odds of hyperuricemia among Iranian adults. This study showed a significant positive association between two indicators of dietary acid load (PRAL, and DAL) and odds of hyperuricemia among Iranian adults. This study showed a significant positive association between two indicators of dietary acid load (PRAL, and DAL) and odds of hyperuricemia among Iranian adults. Querschnittstudie DOI: 10.1007/s11255-023-03876-8 Study: moderate evidence | Seifi N, Bahari H, et al. Higher dietary acid load is associated with the risk of hyperuricemia. Int Urol Nephrol. 2023;56(5):1743–1749. |
| 70. | * Nutrient content and estimated potential renal acid load (PRAL) of 114 frequently consumed foods and beverages (related to 100-g edible portion) Plus eine Tabelle, organisiert nach Food Groups methodische / analytische Modellvalidierungsstudie DOI: 10.1016/S0002-8223(95)00219-7 Study: weak evidence | Remer T, Manz F. Potential renal acid load of foods and its influence on urine pH. J Am Diet Assoc. 1995;95(7):791–797. |
| 71. | * Background: In 1995, Remer and Manz reported potential renal acid load (PRAL) values of various foods, quantifying their estimated impact on acid–base balance. Their estimation considered ionic dissociation, sulphur metabolism and intestinal absorption rates for several micronutrients and proteins. Notably, PRAL values are based on food content data from the early 1990s and may nowadays no longer adequately reflect accurate estimates. Some foods’ macronutrient and mineral content has declined over the past three decades due to changes in soil mineral health. Aim: We hypothesize that the 1995 PRAL values no longer adequately reflect reliable estimates of the current acid–base impact of some foods. Methods: Based on specific examples, we argue that these values overestimate the alkalizing effects of various fruits and vegetables. Conclusion: Discussing evidence in favour of (and against) our hypothesis, we conclude that the 1995 PRAL estimates should nowadays rather be used as a relative guidance and reviewed carefully. The PRAL formula estimates the acid load from food items and considers ionic dissociation, sulphur metabolism and intestinal absorption rates for potassium, phosphate, magnesium, calcium and protein: PRAL (mEq / day) = (0.49 × total protein (g / day)) + (0.037 × phosphorus (mg / day)) − (0.021 × potassium (mg / day)) − (0.026 × magnesium (mg / day)) − (0.013 × calcium (mg / day)) One of the main reasons is that over the past three decades, there has been a substantial decline in the content of essential minerals in some fruits and vegetables. All elements – except phosphorus – declined in their concentrations between the 1940s and 2019 (Mayer et al., 2022). A common example is magnesium content, which declined by approximately 10% in many fruits and vegetables (Mayer et al., 2022). When glancing at the PRAL formula, we encounter that magnesium has a relatively high weighting factor (in comparison to the other minerals): −0.026 (Figure 2). Although of minimal impact in comparison to protein intake, it is noteworthy that when regarded as a whole (e.g. over a period of 24 h), this may have a substantial impact on the total PRAL sum. Going back to PRAL estimations, our hypothesis would have relevant consequences. Based on our hypothesis, the PRAL tables in their current form overestimate the alkalizing effect of some fruits, vegetables and grains. As a corollary, a much higher intake of plant foods would be necessary to compensate for the acidifying properties of meat, dairy and eggs. From a nutrition therapy perspective, a more suitable approach however (particularly in light of the increasing prices for plant foods) would be to reduce the amount of acid-inducing animal foods. Based on our hypothesis, the impact of this measure to reduce PRAL values should be even more valued (also with regard to the fact that protein has the highest weighting factor in PRAL calculations). Another crucial implication of our research is that the 1995 PRAL table should be used with caution when estimating total PRAL values (e.g. for scientific reasons or to guide therapies to a certain desired range). The existing lists should rather be used as a relative guidance (e.g. to indicate whether a certain food has alkalizing or acidifying properties) but not for precise calculations. Hypothesenpapier DOI: 10.1177/0260106023116466 Study: weak evidence | Storz MA, Ronco AL. The 1995 potential renal acid load (PRAL) values may no longer adequately reflect the actual acid–base impact of certain foods: A hypothesis. Nutr Health. 2023;29(3):363–368. |
| 72. | * Table 1. Average potential renal acid loads (PRAL) of certain food groups (related to 100 g edible portion), zitiert als Quelle: 27.Remer T. Influence of diet on acid-base balance. Semin. Dial. 2000;13:221–226. doi: 10.1046/j.1525-139x.2000.00062.x. Jedoch genauso in unserer Quelle 70 vorhanden. Narratives Review DOI: 10.3390/nu10040512 Study: weak evidence | Siener R. Dietary Treatment of Metabolic Acidosis in Chronic Kidney Disease. Nutrients. 2018;10(4):512. |
| 73. | ● Estimated acid load* of frequently consumed foods and drinks (based on 100g). Modified according to Remer and Manz, Journal of the American Dietetic Association 1995; 95:791–797. *PRAL = potential renal acid load based on 100g of the foods, specified in mEq = physical-chemical unit of material. Blue = Alkalinizing Yellow = Acidifying Estimated potential renal acid load (PRAL in mEq/100g) of frequently consumed foods and drinks (based on 100g). Modified according to Remer and Manz. Website | Pascoe ca: Food Table - PRAL Values. |
| 74. | ● Geschätzte Säurebelastung von häufig verzehrten Nahrungsmitteln und Getränken (bezogen auf 100 g). PRAL = potential renal acid load = potentielle, die Nieren betreffende Säurelast, bezogen auf 100 g des Nahrungsmittels, angegeben in mEq = physikalisch-chemische Stoffmengeneinheit. Die alte Klassifizierung von Nahrungsmitteln nach ihrem PH-Wert wird heute, nach neuesten wissenschaftlichen Erkenntnissen, durch den sogenannten PRAL-Wert ersetzt. PRAL steht für die „Potential renal acid load" und wird in Milliäquivalent pro 100 g Lebensmittel (mEq/100 g) angegeben. Der Wert gibt also genauen Aufschluss darüber, wie hoch die Säureausscheidung über die Niere beim Verzehr von 100 g eines Lebensmittels ist. Werte mit einem negativen Vorzeichen (Farbe grün) stehen für eine „basische“ Verstoffwechselung, Werte mit positiven Vorzeichen (Farbe rot) für eine saure Verstoffwechselung. Ein optimaler Säure-Basen-Haushalt besteht aus 70-80% basenbildenden und 20-30% säurebildenden Nahrungsmitteln. PRAL-Werte geben den Grad der Säurebelastung an: hoher negativer Wert = sehr basisch, hoher positiver Wert = stark säuernd Blau = basisch verstoffwechselte Nahrungsmittel Gelb = sauer verstoffwechselte Nahrungsmittel Quellenangabe bei Burgerstein: Der PRAL-Wert wurde von Dr. Thomas Remer und Dr. Friedrich Manz entwickelt. 1995 veröffentlichten die beiden Professoren die PRAL-Tabellenwerte für eine Vielzahl von Lebensmitteln. Website | Burgerstein at: Nahrungsmitteltabelle – PRAL-Werte. |
| 75. | * Querschnittstudie DOI: 10.1080/07315724.1992.10718238 Study: moderate evidence | Brulé D, Sarwar G, Savoie L. Changes in serum and urinary uric acid levels in normal human subjects fed purine-rich foods containing different amounts of adenine and hypoxanthine. Journal of the American College of Nutrition. 1992;11(3):353–358. |
| Wir haben Studien und Bücher zu Ernährung und Gesundheit nach folgenden 3 Evidenz-Kategorien markiert: grün=starke Beweiskraft, gelb=mittlere, violett=schwache. Die restlichen Quellen sind grau markiert. Eine ausführliche Erklärung finden Sie in unserem Beitrag: Wissenschaft oder Glaube? So prüfen Sie Publikationen. | ||

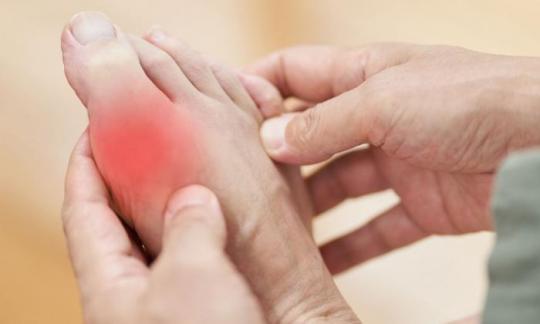
Kommentare