Phenylalanine (Phe, F)
Did you know that phenylalanine is not only important for protein production, but also serves as a precursor for neurotransmitters and the skin pigment melanin? Discover with us which natural foods are particularly rich in this essential amino acid, etc.
A balanced, plant-based diet with few to no industrially processed foods generally provides sufficient macro- and micronutrients, with the exception of vitamin B12. However, phytochemicals are particularly relevant for maintaining health and healing, even though they are not considered essential nutrients – apart from vitamins.
---
Phenylalanine
(Phe, F) is one of eight (ten in children, including histidine and arginine) amino acids considered essential (non-dispensable) for the human body. Humans use 21 proteinogenic amino acids. These are the building blocks of proteins. With the exception of two (lysine and threonine), the body can adapt protein production to its needs, even though eight are traditionally considered essential.
Occurrence
Legumes, in particular, contain high levels of phenylalanine, such as soybeans (2122 mg/100 g) and kidney beans (1220 mg/100 g).1
| Groceries | Protein g/100 g (USDA) | Phenylalanine mg/100 g (USDA) | % of phenylalanine in total protein |
| 36 g | 2122 mg | 5.8% | |
| 30 g | 1733 mg | 5.7% | |
| 25 g | 1132 mg | 4.6% | |
| 23 g | 1130 mg | 5.1% | |
| 15 g | 711 mg | 4.7% | |
| Whole wheat flour | 14 g | 646 mg | 4.7% |
| 8 g | 410 mg | 5.2% |
Nuts, such as peanuts (1400 mg/100 g) and almonds (1130 mg), as well as seeds like flaxseed (960 mg) and sesame seeds (940 mg), are very good sources of phenylalanine. Various whole grains also provide high amounts of this amino acid. Vegetables and berries contain roughly the same amount, while fruits contain significantly less.1
Meat and fish, as well as eggs (680 mg) and milk (460 mg), contain less phenylalanine than soybeans, pumpkin seeds, chickpeas or almonds.
Storage and Preparation Losses
Heating food denatures proteins, thus altering their properties. A fried egg is a good example; it denatures due to the heat in the pan. When an egg is heated, the liquid yolk and the egg white, the main protein source, solidify. The proteins in the egg thicken, a process known as "coagulation" or "flocculation". The egg white coagulates at 60 °C, the yolk at 65 °C, and complete coagulation occurs at 70 °C.
Meat proteins begin to denature during cooking at 40–60 °C or 80 °C. The oxidation of amino acids by reactive oxygen can severely impair their function.
Nutrition - Health
Phenylalanine, which makes up more than half of the aspartame molecule, is an essential component of body proteins and an important precursor to many aromatic compounds necessary for normal bodily function.7
Essential means that our body cannot produce (synthesize) these amino acids itself, but needs them in certain amounts to maintain growth and nitrogen balance. 10
It is now known that the body only irreversibly transaminates lysine and threonine, and that only these two are truly essential. On average, vegans consume about twice the amount of threonine and are well-supplied with lysine. Specific dietary habits, such as fruitarianism or the 80/10/10 diet, or even more extreme ones, can lead to deficiencies over time. This can occur for extended periods without any immediately noticeable symptoms.
| This is not just for vegans or vegetarians: Vegans often eat unhealthily. Avoidable nutritional mistakes. |
Long-term daily requirement
The body produces the amino acid tyrosine from phenylalanine.3,5 Therefore, the need for phenylalanine depends on the tyrosine content of the diet. The estimated average minimum requirement for phenylalanine when there is an excess of tyrosine is 9,1 mg/1 kg body weight per day. On a tyrosine-free diet, the average maximum phenylalanine requirement for healthy adults is 42 mg per 1 kg body weight per day. Optimal utilization of the two amino acids occurs when the phenylalanine to tyrosine ratio in the diet is 60:40. Sometimes, information is also given for the combined requirement (phenylalanine and tyrosine). This is 14 mg per kg body weight per day.5
According to scientific tables (Geigy, Vol. 2, p. 232), the daily requirement for phenylalanine and tyrosine combined is 16 mg/kg body weight. This includes a 30 % allowance for individual variations. For a person weighing 75 kg, this would be 1,2 g/day. More recent studies recommend even lower amounts.
If tyrosine is present in excess, we need approximately 9,1 mg of phenylalanine per kg of body weight per day. Optimal utilization of the two amino acids occurs at a phenylalanine to tyrosine ratio of 60:40.5
Deficiency symptoms
A phenylalanine deficiency rarely occurs with a balanced diet. A natural vegan diet more than adequately covers phenylalanine requirements. Infants are particularly at risk if they are weaned from breast milk too early and receive inappropriate nutrition.
A deficiency in tyrosine and phenylalanine caused by a manipulated diet can reduce dopamine synthesis in the brain and thus impair dopamine activity in the brains of healthy individuals.13
A prolonged deficiency can lead to reduced performance, decreased appetite, and metabolic disorders. The diseases kwashiorkor and marasmus, which can occur with very long-term protein and general energy deficiency, are now rarely found in the Western world.10
Oversupply
Elevated phenylalanine levels occur when its breakdown or conversion into other substances is impaired. This is the case in the genetically determined metabolic disorder phenylketonuria (PKU), also known as phenylalanine hydroxylase (PAH) deficiency. This autosomal recessive disorder prevents the complete breakdown of phenylalanine and leads to an accumulation of phenylalanine in the blood.
Left untreated, this can lead to severe intellectual disability, epilepsy, and behavioral disorders. The prevalence is approximately 1 in 10,000 newborns worldwide. People with phenylketonuria must adhere to a low-phenylalanine diet for life.
A statement printed on food packaging stating "contains a source of phenylalanine" indicates the presence of the sweetener aspartame, which releases phenylalanine during digestion.6
In chronic kidney failure, the body cannot efficiently convert phenylalanine to tyrosine. Therefore, tyrosine and the tyrosine-to-phenylalanine ratio are lower in the blood and many tissues, while phenylalanine levels are normal or slightly elevated.8 For this reason, a low-protein diet is recommended for individuals with impaired kidney function to prevent increased production and accumulation of urea in the kidneys due to amino acid breakdown. The book * The China Study* vividly demonstrates, with strong evidence, that we typically suffer from an excess, not a deficiency, of protein (see detailed book review): Only in recent years have scientists discovered that animal protein, once considered the highest quality protein (biological value), does not promote optimal health, but rather the opposite.
Functions in the body
Phenylalanine has the following functions in the body:2,3,9,12
- Phenylalanine and, dependent on it, tyrosine contribute to the synthesis of important endogenous proteins, such as the neurotransmitters adrenaline, noradrenaline, dopamine, and serotonin, as well as the skin pigment melanin and the hormone thyroxine (T4). Phenylalanine can improve memory and suppress appetite.
- Unlike other amino acids, phenylalanine has special functions in the body not only in its natural L-form but also in its D-form. Medicine uses synthetically produced D- and L-phenylalanine (DLPA) as pain relievers or to treat depression. The antidepressant effect of L-phenylalanine may be due to its role as a precursor in the synthesis of the neurotransmitters norepinephrine and dopamine.
Absorption and Metabolism
The digestion of ingested proteins begins in the stomach with the action of pepsin. Pepsins break down the proteins into peptides. Further breakdown into individual amino acids occurs in the small intestine through the action of peptidases. The liver converts phenylalanine into the non-essential amino acid tyrosine when dietary tyrosine intake is low. With a tyrosine-deficient diet, the body can convert up to half of the ingested phenylalanine into tyrosine. Conversely, with a tyrosine-rich diet, the body's need for phenylalanine decreases by 50 %.2,9
Storage - Consumption - Losses
70-80% of free amino acids are found in skeletal muscle, a smaller proportion in blood plasma. The liver is the central organ for amino acid regulation. Breakdown also occurs there, e.g., through deamination and conversion to urea, to prevent cytotoxic effects.3
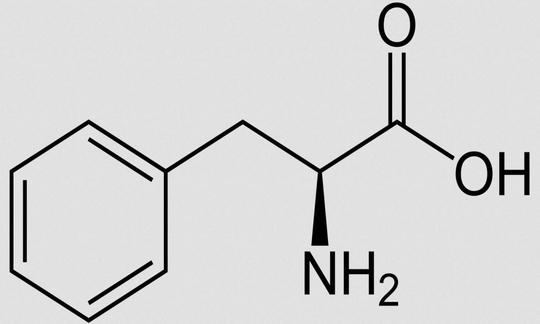
Phenylalanine is a chiral, aromatic α-amino acid with a hydrophobic side chain.
Structure
Phenylalanine is a chiral, aromatic α-amino acid with a hydrophobic side chain. Structurally, phenylalanine is derived from alanine and is amphiphilic. Other names for L-phenylalanine: 2-Amino-3-phenylpropionic acid; 2-Amino-3-phenylpropanoic acid. Molecular formula: C9H11NO2. Abbreviations: Phe, F (one-letter code).12
Bibliography - 14 Sources (Link to the evidence)
| 1. | US-Amerikanische Nährwertdatenbank USDA. |
| 2. | Kasper H, Burghardt W. Ernährungsmedizin und Diätetik. 11. Auflage. Elsevier GmbH. Urban & Fischer Verlag: München. 2009. |
| 3. | Biesalski HK, Grimm P. Taschenatlas der Ernährung. 6. Auflage. Georg Thieme Verlag: Stuttgart und New York. 2015. |
| 4. | WHO World Health Organization. Protein and amino acid requirements in human nutrition. Technical Report Series: 935. 2002. |
| 5. | Pencharz PB, Hsu JW et al. Aromatic amino acid requirements in healthy human subjects. J Nutr. 2007 Jun;137(6 Suppl 1):1576S-1578S; discussion 1597S-1598S. |
| 6. | Van Spronsen FJ, Blau N et al. Phenylketonuria. Nat Rev Dis Primers. 2021 May 20;7(1):36. |
| 7. | Harper AE. Phenylalanine metabolism. Aspartame. 2nd ed. CRC Press, 2020. 77-109. |
| 8. | Kopple JD. Phenylalanine and tyrosine metabolism in chronic kidney failure. J Nutr. 2007 Jun;137(6 Suppl 1):1586S-1590S; discussion 1597S-1598S. |
| 9. | Litwack G. Metabolism of Amino Acids. Human Biochemistry. 2018. |
| 10. | Watford M, Wu G. Protein. Adv Nutr. 2011 Jan;2(1):62-63. |
| 11. | Ishiwatari N, Fukuoka M et al. Effect of protein denaturation degree on texture and water state of cooked meat. J Food Eng. 2013;117(3):361-369. |
| 12. | Pubchem.ncbi.nlm.nih.gov. Phenylalanine. |
| 13. | Montgomery AJ, McTavish SF et al. Reduction of brain dopamine concentration with dietary tyrosine plus phenylalanine depletion: an [11C]raclopride PET study. Am J Psychiatry. 2003 Oct;160(10):1887-1889. |
| 14. | Ifst Institute of Food Science & Technology. Protein: Coagulation. 2017. |
For this dish, the beans are first soaked in water overnight. In the cookbook, you will find a dandelion salad that complements the recipe well.
The Indian Toor Dal with pigeon peas, cumin and chili is quick and easy to make.
This "Tailgate Chili with Borlotti Beans" is a hearty dish that you can easily modify as you like. Here with corn on the cob and tomatoes.
The cyanobacteria Spirulina (powder etc.) can practically never be bought raw. Because of their blue pigment, they were previously classified as blue-green alga
Soya granules consist of ground, defatted and heated soya beans - it is never raw! Due to its protein content and consistency, it is considered an (organic?) me
Brewer's yeast has a spicy, slightly cheese-like taste. It is therefore used for seasoning or as a cheese substitute in dishes. Arguments for organic.






Comments