Nutritional Programming
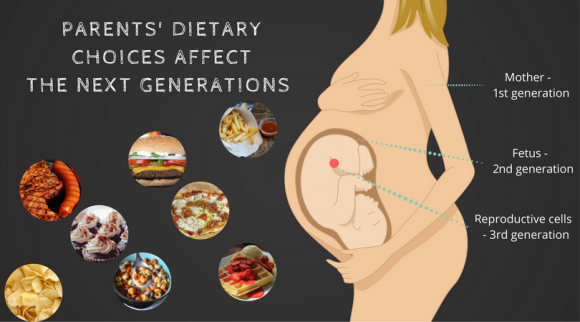
Nutritional programming: how your dietary choices affect future generations.
Table of contents
- Conclusions
- 1. Parents’ dietary choices have an impact on the health and well-being of their children
- 2. Why does this happen?
- 3. Nutritional (metabolic) programming
- 4. What happens on the cellular level
- 5. Epigenetics — the main process that contributes to nutritional programming
- 6. How your lifestyle choices could impact the health of your children
Conclusions
Nutritional programming explains why some people have a predisposition for certain diseases in adulthood. These diseases can occur because of altered development during the fetal stage and early infancy.
According to the “Developmental Origins of Health and Disease” paradigm, the conditions during periconception (2–3 months before getting pregnant), pregnancy, and lactation strongly influence the developing child. There is substantial evidence showing that an unfavorable environment during these periods creates a predisposition to certain diseases, mostly inflammatory, that manifest later in adult life.1
Overnutrition and undernutrition are among the most important elements that can contribute to a harmful environment for the unborn child. These have been directly correlated with metabolic conditions such as hypertension, "insulinresistance", hyperlipemia, and abdominal obesity.2
When the environment is harmful, the embryo, fetus, or newborn adjusts so that it can survive in spite of the detrimental conditions. These adjustments often involve changes in body functions, tissue remodeling, and the activation or silencing of certain genes. Such changes can become permanent and remain active even when the harmful stimuli disappear.
For example, undernutrition of the mother during pregnancy and early infancy can cause children to have an increased risk of developing obesity, diabetes, metabolic syndrome, and cardiovascular diseases later in life. This increased risk is a direct result of being exposed to unhealthy diets and periods of low physical activity.3
A small glossary of terms
Embryo: After the egg is fertilized by the sperm (zygote), it starts dividing and forms a hollow ball of cells (blastocyst) and then moves to the uterus. Here, some of these cells form the placenta while others develop into the embryo. At this stage, the body structure and most of the internal organs are formed. The brain and spinal cord continue to develop throughout the entire pregnancy. New brain cells develop both during the pregnancy and in the first year after birth.
Fetus: The embryo is considered a fetus from the end of the eighth week after fertilization (10 weeks of pregnancy) until birth.
Placenta: The organ that develops in the uterus of a pregnant women and connects the fetus to the mother’s body. The placenta makes the transfer of nutrients and oxygen possible. It also helps remove waste products from the child’s blood and fights against internal infections.
Gestation: This is the period between conception and birth when the embryo and fetus develop in the womb.
1. Parents’ dietary choices have an impact on the health and well-being of their children
It is common knowledge that nutrition and lifestyle have a direct impact on our personal health.
New studies3 provide considerable evidence showing that our health and well-being is determined not only by our own personal habits of diet and physical activity, but also by
- the environment in which we developed during the fetal stage (e.g., access to nutrients, mother’s hormonal status and stress level, and placenta characteristics); and
- the dietary choices our parents make.
In other words, poor lifestyle habits not only affect us, but also our children.
In the short term, all of these changes influence the child’s brain development, "growing", body composition, and metabolism. In the long term, they can also influence the child’s cognitive capacity, immunity, and energy level, which can affect (among other factors) school performance, the education level acquired, a person’s capacity to handle a job, and overall health.5
The same idea is supported by “The Developmental Origins of Health and Disease” (DOHaD) paradigm, developed by Barker in 1990. It states that conditions during periconception, gestation, and lactation shape the developing child and in the case of an unfavorable environment can lead to a predisposition for adult-onset diseases.1
2. Why does this happen?
Starting in the earliest stages of our development (embryonal and fetus stages), we are able to respond to environmental triggers through adjustments at the cellular, molecular, and biochemical levels. If certain nutritional stressors or stimuli induce such adaptations, the changes in the body continue to exist and be expressed, even in the absence of the stimuli that initiated them.6
Studies on animals have focused on two major scenarios:
- overnutrition of the mother — through a diet high in fat, cholesterol, and protein, all of which can lead to maternal obesity
- undernutrition of the mother — through a deficiency of micronutrients (e.g., Ca, Fe, Na, and Zn) and a lack of macronutrients (e.g., protein).
One research study showed that when animals with a low birth weight ate a high-calorie diet during their adult life, they had a tendency to gain fat. This was because their bodies were programmed to restrict the energy expenditure as an adaptive response to the deficiencies in the fetal period.7
The result of another study6 showed that a low-protein diet and caloric restriction during pregnancy can cause alterations in fetuses’ pancreatic islets (e.g., reduced vascularization and a reduced capacity to produce beta cells) and decreased sensitivity to insulinum in muscles. Even if the baby is fed a normal diet after birth and during childhood, the changes in the organs are irreversible and increase the baby’s predisposition for diabetes and metabolic diseases.
Given the fact that plasticity (the capacity of cells to transform into other different cells) is one of the main characteristics of cells during the embryonic and fetal periods and that the internal organs are in the process of development and structuring, the impact of any disease-promoting factor is stronger in this period than at any time later in life.
3. Nutritional (metabolic) programming
Nutritional (metabolic) programing is the process through which the quality and quantity of the nutrients consumed during pregnancy permanently impact the developing fetus.
To date, research studies and experiments have been developed only on animals (rats and sheep), by overfeeding them or restricting specific nutrients and observing the impact on their offspring. On humans, only epidemiologic studies have been possible; conclusions have been drawn based on observing correlations that exist between conditions during pregnancy and health problems people develop later in adult life.
Why nutritional programming can’t be proved in experiments on humans
Nutritional programming occurs mostly in the embryonal and fetus stages, when the degree of cell plasticity is the highest. To study embryonic cells and the impact of the environment upon them, scientists would have to create or generate these cells, most likely by in vitro fertilization, a process which raises many ethical, moral, religious, and legal concerns.
Most of the experimental research studies on animals, which modify external conditions and then study the impact on the newborn and adult animal, can’t be replicated on humans.
The reasons8 why animal studies are still considered relevant in this field are as follows:
- Rats, mice, and humans have a 99 % gene similarity.
- The environment can be easily controlled.
- The life cycle is short, which makes it possible to trace the modifications.
The quality and quantity of nutrients consumed by the mother during pregnancy can lead to significant changes in the offspring regarding the structure of the brain, kidneys, and heart. Correlations have also been established that show an increased risk of coronary heart disease and disorders related to "insulinresistance".7
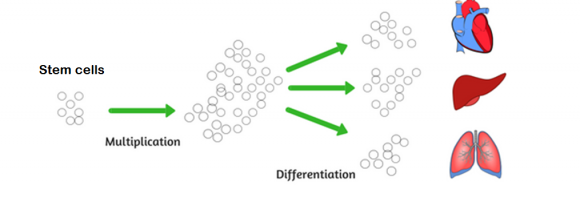
4. What happens on the cellular level
Human development in the uterus is based on two main processes: cell multiplication and cell differentiation (specialization). Initially, there is a small pool of cells (stem cells) capable of becoming any organ or tissue in the body. These cells multiply and then specialize, taking on the characteristics of a special organ. The time when these processes take place differs depending on the organ. The heart, for example, takes form earlier than the sex organs, and the lungs develop only in the final weeks of pregnancy.
The nutritional variations in a mother’s diet can influence the multiplication or differentiation process. Adverse conditions during proliferation can cause organs to be smaller, even though they have normal cell characteristics. And adverse conditions during differentiation can result in normal-sized tissue having an altered profile and functionality.7
The results of a number of research studies have shown that in most cases poor maternal nutrition during pregnancy leads to changes on the following levels:
- Kidneys: animals exposed to nutrient deficits during the fetal stage have 30–40 % fewer nephrons than normal.7 In humans, studies on the Australian aboriginal population showed a 20 % higher renal failure rate as compared to the Caucasian population.9
- Brain: research studies on rats have shown that undernutrition reduces the density of capillaries in the brain and that the density of neurons responsible for appetite regulation was lower.10
- Pancreas: due to decreased nutritional supply, the pancreatic cells in animals have fewer islets of Langerhans and the blood supply is reduced.7
Is fetal "growing" a reliable measurement for the future health of a baby?
Up to the present, fetal "growing" (weight and length) has been one of the most researched topics used to show the association between environmental conditions, fetus development, and health conditions in later life.
The research studies published by Barker and his colleagues, after observing thousands of people born over a period of twenty years, have established that a low birth weight is a strong predictor for high blood pressure, a higher blood clotting risk, impaired glucose tolerance, type 2 diabetes, and metabolic syndrome.7
Other factors that predict future health conditions include a reduced abdominal circumference, a large head circumference in relation to the body length, and low weight in relation to body length.
A research study conducted by Rosenboom11 regarding the Dutch famine period and its impact on the children born during that time clearly shows that malnutrition during the gestational period affects adults’ health in later life.
Research studies on animals have established that the impact of these environmental conditions goes beyond fetal "growing". Yet, because this impact is less evident and expressed on various levels, it is difficult to observe in experimental studies.
Even if the fetus continues to grow normally, the nutrient deficit can have a significant impact on its future health. From this perspective, birth weight alone cannot be the only predictor for health during adulthood.
After studying the children born during and after the Dutch famine period, Rosenboom’s concluded that maternal malnutrition during gestation may permanently affect adult health, without affecting the size of the baby at birth.
10 Furthermore, the effects of undernutrition depend upon timing during gestation and the specific organs and systems developing during this critical time window.
For example, those individuals conceived before the Dutch famine and exposed to an energy-poor fetal environment late in gestation then as adults had an increased risk for "insulinresistance" and impaired glucose tolerance. And those conceived during the famine had an increased risk for high serum cholesterol and coronary heart disease.11
Variations in a mother’s nutrition are the best predictor for fetal changes, and they are also the strongest motor for fetal programming. However, there are many other factors that can simultaneously influence the fetus’s nutritional status. These include the mother’s body composition, age, physical activity, blood flow to the placenta, the efficiency of nutrient transfer, and the mother’s hormonal balance.
5. Epigenetics — the main process that contributes to nutritional programming
A mother’s nutrition exerts its influence upon the developing fetus through changes in the process of cellular multiplication and differentiation, but also by affecting the fetus’ genes expression (epigenetic processes).
How epigenetics works
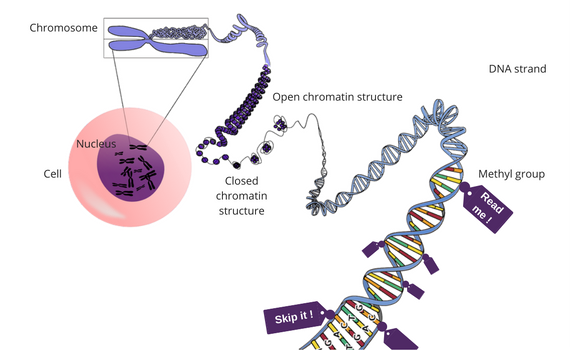
Imagine the human body is like a huge and complex factory, with the body cells being the fundamental working units. In order to know how to function, the cells need information, basic rules, and procedures. This huge database of rules and procedures is found in the DNA and carefully organized in specific sequences of information called genes. There are over 20'000 genes, and each cell has access to all of them, but not all of this information is “read” and implemented. The information is accessed and applied selectively, according to additional instructions, called epigenetic markers. All of these epigenetic markers together form the epigenome, which is distinctive for every person and is strongly influenced by the environment. Read more here about the difference between genetics and epigenetics.
Epigenetics (literally translated “above genetics”) is the process of highlighting or defining certain parts of these rules and procedures, making them “actively” visible, telling cells to pay attention to and to express those sections. Other epigenetic factors silence certain genes, making them inaccessible, so that the cells cannot read and apply the information.
Primary mechanisms of epigenetic modification
The primary mechanisms involved in epigenetic modification are12:
- Methylation — a methyl group (chemical compound) attaches to the DNA and modifies the function of a specific gene.
- Histone modification — since the DNA is extremely long as compared to the size of the cell nucleus, it needs to be wrapped around protein structures called “histones” in order to fit in. And in this way, it forms a chromatin structure. When this wrap is really tight, the information in the sequence cannot be read by the cell. If, on the contrary, the ties between the histones become loose, the information can be read and applied.
For example, animals in one study were fed a diet deficient in methyl donors, folic acid, vitamin B12, and methionine for a period of 8 weeks prior to conception and for the first 6 days of pregnancy. The male offspring from these ewes were "insulinresistant" and had elevated blood pressure.13
This phenomenon is the most visible in identical twins who spend their lives in different environments. Even if the genome (all the hereditary information) is the same, the two people end up being different in terms of body functionality and health outcomes.
Epigenetics — the good and the bad
The role of epigenetics consists of helping the body to accommodate to the environment and life major changes. For example, the primary mechanisms of epigenetics help us survive during famine periods, the female body to change during pregnancy, and adolescents’ bodies as they change during puberty.
The problems occur when these epigenetic changes start to generate diseases. Sometimes, the genes that are made active are the genes associated with certain diseases. Cancer, for example, can be caused if too many "growthpromoting" genes are activated or, on the contrary, if tumor-suppressing genes are silenced.
Epigenetic modifications can be transmitted by genes
An important thing to take into consideration is the fact that these epigenetic markers aren’t fixed. Under the influence of nutrition and the environment, they can change throughout a person’s life, and some of them can even be inherited from previous generations and transmitted further.
In an experiment conducted by the Center for Reproductive Biology at Washington State University, pregnant rats were exposed to relatively high levels of methoxychlor (insecticide) and vinclozolin (fungicide). The male rats in the next generation then exhibited decreased sperm production and increased infertility. The research study proved that the cause of these manifestations was a methylation process that affected two genes. Without being exposed to pesticide substances anymore, the next four generations were observed. A total of 90% of the male rats showed the same adverse effects.14
Research studies developed by Dr. Erica Watson at the University of Cambridge showed that when either the maternal grandmother or the maternal grandfather had a specific genetic mutation (known as Mtrr mutation) which causes folic acid deficiency, their genetically normal grandchildren who did not inherit the genetic mutation were still at risk of certain developmental abnormalities.1
6. How your lifestyle choices could impact the health of your children
For a long time, scientists thought that undernutrition and Western diseases were mutually exclusive. Surprisingly, the experiments that restricted the nutritional intake of pregnant animals generated the same results in the offspring as did the experiments where the animals were overnourished (e.g., high blood pressure, glucose intolerance, "insulinresistance", and a greater predisposition toward obesity.17
Here are some of the main effects the parents’ nutrition, weight status, and lifestyle prior and after conception might have on their children:
| Parental Nutrition and Lifestyle | Possible Impact on Children |
| High-fat diet | Elevated blood pressure, vascular diseases (Endothelial dysfunction)18 |
| High-protein diet | Alteration of energy consumption, obesity19 |
| High cholesterol level in early pregnancy (maternal) | Overweight/obesity, increased fat mass, increased cholesterol level at age four23 |
| Undernutrition during pregnancy (maternal) | Increased production of glucose in the liver, impaired oxidation of fatty acids, resistance to ketosis, changes in glucose uptake2 |
| Overnutrition (maternal) | Obesity, glucose intolerance, altered brain appetite regulators |
| Low-protein diet | The expression of 102 genes in the hypothalamus and of 36 genes in the kidney is modified.20 |
| Obesity before and during pregnancy and high leptin levels due to obesity (maternal) | High leptin levels during the period in which the fetus’ hypothalamus develops lead to leptin resistance, hyperphagia, and a diminished capacity of the future child to regulate its appetite.21 Higher blood pressure, greater adiposity, and "insulinresistance".1 |
| Excess weight before pregnancy (maternal) | Increased fat mass and high levels of systolic pressure in the future child22 |
| Diet low in iron prior and during pregnancy (maternal) | Changes in cardiac development during the fetal period may lead to elevated blood pressure and changes in fatty acid metabolism.7 |
| Started smoking at an early age (paternal) | Higher body mass index of sons23 |
| Alcohol consumption prior to conception (paternal) | Reduced birth weight, developmental retardation, compromised immunity, behavioral deficits, hyperactivity, and reduced cognitive performance24 |
The severity of these effects is related to the duration and quantity of the exposure to the harmful substance, as well as to the developmental period when the harmful influence occurred.
Until recently, scientists believed that epigenetic markers impacting the fetus’ genes would have a permanent effect on the health and well-being of an individual throughout their entire life. New emerging research studies are investigating whether these epigenetic markers might instead remain flexible during adulthood, which could lead to positive changes when environmental conditions become favorable.
Each person shares responsibility for their own genes. Gottfried Schatz
In other words, even if certain epigenetic modifications took place during the fetal stage and lead to health conditions in adulthood, there might still be a possibility to alter and reverse the action of these epigenetic markers as adults. The constant exposure to beneficial and positive environmental stimuli (e.g., healthy nutrition and lifestyle and positive emotions) might change the activation or the silencing of the genes, preventing or reversing chronic diseases such as cancer, diabetes, hypertension, and heart problems.
At this stage, most of this information is still speculative. However, many new research studies are focusing on the impact that parents’ nutrition and lifestyle choices have on children through the processes of epigenetic modifications and nutritional programming.
Literature — sources:
24 sources
- Tarrade A, Panchenko P, Junien C, Gabory A. Placental contribution to nutritional programming of health and diseases: epigenetics and sexual dimorphism. Journal of Experimental Biology 2015.
- Martínez JA, Cordero P, Campión J, Milagro FI. Interplay of early life nutritional programming on obesity, inflammation and epigenetic outcomes. Proc Nutr Soc. 2012 May;71(2):276-83.
- Wadhwa PD, Buss C, Entringer S, Swanson JM. Developmental Origins of Health and Disease: Brief History of the Approach and Current Focus on Epigenetic Mechanisms. Seminars in reproductive medicine. 2009;27(5):358-368. doi:10.1055/s-0029-1237424.
- Vickers, MV. Early Life Nutrition, Epigenetics and Programming of Later Life Disease. Nutrients 2014.
- Uauy R, Kain J, Corvalan C. How can the Developmental Origins of Health and Disease (DOHaD) hypothesis contribute to improving health in developing countries? The American journal of clinical nutrition. 2011.
- Patel MS, Srinivasan M. Metabolic Programming: Causes and Consequences. The Journal of Biological Chemistry, January 2011; 277.
- Langley-Evans SC. Nutritional programming of disease: unravelling the mechanism. Journal of Anatomy. 2009;215(1):36-51.
- Gunter C, Dhand R. Human biology by proxy. Nature 420, 509, 5 December 2002.
- Singh GR, Hoy WE. Kidney volume, blood pressure, and albuminuria: findings in an Australian aboriginal community. J Kidney Dis. 2004.
- Plagemann A, Harder T, Rake A, et all. Hypothalamic nuclei are malformed in weanling offspring of low protein malnourished rat dams. J Nutr. 2000.
- Roseboom TJ, van der Meulen JH, Ravelli AC, et all. Effects of prenatal exposure to the Dutch famine on adult disease in later life: an overview. Mol Cell Endocrinol. 2001.
- Kanherkar RR, Bhatia-Dey N, Csoka AB. Epigenetics across the human lifespan. Front Cell Dev Biol. 2014; 2: 49.
- Sinclair KD, Allegrucci C, Singh R, et al. DNA methylation, "insulinresistance", and blood pressure in offspring determined by maternal periconceptional B vitamin and methionine status. Proc Natl Acad Sci USA. 2007;104:19351–19356.
- Weinhold B. Epigenetics: The Science of Change. Environmental Health Perspectives. 2006.
- Anway MD1, Cupp AS, Uzumcu M, Skinner MK. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science. 2005 Jun 3;308(5727):1466-9.
- Padmanabhan N, Jia D, Geary-Joo C, Wu , Ferguson-Smith AC, Fung E. et all. Mutation in Folate Metabolism Causes Epigenetic Instability and Transgenerational Effects on Development. J. Cell, 2013 Sept. 2.
- Langley-Evans SC. Developmental programming of health and disease. Proc Nutr Soc. 2006.
- Khan IY, Taylor PD, Dekou V et all. Gender-linked hypertension in offspring of lard-fed pregnant rats. Hypertension. 2003.
- Daenzer M, Ortmann S, Klaus S, Metges CC. Prenatal high protein exposure decreases energy expenditure and increases adiposity in young rats. J Nutr. 2002.
- Langley-Evans SC, Bellinger L, McMullen S. Animal models of programming: early life influences on appetite and feeding behaviour. Matern Child Nutr. 2005.
- Samuelsson AM, Matthews PA, Argenton M, Christie MR, McConnell JM, Jansen EHJM et all. Diet-Induced Obesity in Female Mice Leads to Offspring Hyperphagia, Adiposity, Hypertension, and "InsulinResistance". A Novel Murine Model of Developmental Programming. Hypertension. 2008;51:383-392.
- Daraki V, Georgiou V, Chalkiadaki G, Karachaliou M, Koinaki S. Metabolic dysregulation in early pregnancy in association with offspring cardiometabolic risk in preschool children. Program of prevention and early diagnostic of obesity and neurodevelopment disorders in preschool age children, Crete, Greece, NSRF 2007-2013 project, MIS 349580.
- Pembrey ME, Bygren LO, Kaati G, Edvinsson S, Northstone K, Sjöström M, Golding J, ALSPAC Study Team. Sex-specific, male-line transgenerational responses in humans. Eur J Hum Genet. 2006 Feb; 14(2):159-66.
- Curley JP, Mashoodh R, Champagne FA. Epigenetics and the Origins of Paternal Effects. Hormones and behavior. 2011;59(3):306-314.


Comments